10.4: Synaptic Mechanisms of Learning and Memory
- Page ID
- 118707
This page is a draft and under active development. Please forward any questions, comments, and/or feedback to the ASCCC OERI (oeri@asccc.org).
Learning Objectives
- Describe Hebb's theory of the engram
- Define Hebb's rule
- Define Hebb synapses and cell assemblies
- Describe Kandel's findings about the synaptic changes mediating habituation and sensitization in the sea slug, Aplysia
- Explain the dual-trace theory of memory
- Describe the role of the hippocampus in learning and memory
- Describe the role of the cerebellum and amygdala in learning and memory
- Discuss Long-term Potentiation (LTP) and Long-term depression and learning and memory
- Describe changes in dendritic spines associated with learning and memory
- Discuss how LTP and anatomical changes in synaptic spines might be related to Hebb's dual-trace theory
Overview
Donald Hebb's (1949) dual-trace theory of the physical basis of memory focused on changes at the synapse as the basis for learning and memory. This influential theory led to the discovery of changes in transmitter release associated with learning in the invertebrate nervous system of the sea slug. Other researchers found sustained increases in synaptic conductance caused by high frequency stimulation of the pre-synaptic neuron (Long-term Potentiation, LTP) as well as sustained decrease in synaptic strength known as Long-term Depression (LTD). Researchers have also discovered anatomical changes in dendritic spines associated with LTP and learning and memory, suggesting anatomical changes at synapses as a possible mechanism for learning and long-term memory at the synaptic level. Although early studies in search of the "engram", the physical basis for learning and memory, concluded that these functions were widely distributed throughout the brain, later research found that the hippocampus is critical for formation of long-term explicit memories and that the cerebellum is involved in implicit memory, whle the amygdala plays an essential role in emotional memories.
The Search for Learning and Memory in the Synapse
by Kenneth A. Koenigshofer, Ph.D.
Just as information is stored on digital media such as DVDs, hard drives, and flash drives, the information in our long-term memory must be physically stored in the brain. According to current theory, the ability to maintain information in long-term memory involves a gradual strengthening of the synaptic connections among neurons. When pathways in these neural networks are frequently and repeatedly activated, the synapses become more efficient permitting enhanced communication among neurons in the network, and these changes create memory (Saylor Foundation, 2012). As we will see, this view of the physical basis of memory was heavily influenced by the ideas of a Harvard biological psychologist named Donald Hebb.
Over a century ago a Russian physiologist named Ivan Pavlov proposed a theory of how learning occurred in the brain. As you recall from from module 10.2, Pavlov discovered classical conditioning when he observed that repeated pairings of a bell and food eventually led dogs to salivate at the sound of the bell, even when food was not present. To explain this phenomenon, Pavlov hypothesized that the conditioned stimulus (CS), a bell in this case, generated a locus of neural activity in sensory cortex (auditory cortex in this experiment) which radiated outward over the cortical surface. This was followed by a similar locus of cortical neural activity generated by the unconditioned stimulus (US) (meat powder in Pavlov's classic experiments) which also set up waves of radiating neural activity. Pavlov proposed that these two expanding fields of neural activity, originating from different areas of cortex, would intersect one another. According to Pavlov, this intersection of the cortical fields of neural activity, generated by the CS and US, formed the neurological basis for the association between CS and US.
This early theory of the physical basis of learning and memory was put to the test by experimental psychologist, Karl Lashley, of Harvard and then later, the Yerkes Laboratory of Primate Biology. Lashley made crisscross cuts throughout the cerebral cortex of rats and then attempted to condition them. He did this to disrupt any radiating neural activity in the cortex that might be present during conditioning, as Pavlov had proposed. Lashley found that the rats still could be conditioned and that they retained the conditioned response later in spite of the crisscross cuts over their entire cerebral cortex. This disproved Pavlov's theory that the physical basis of learning an association by classical conditioning was the intersection of waves of neural activity radiating outward from cortical loci.
Lashley, like Pavlov, was interested in finding the "engram." This term refers to the physical memory trace, the neural representation of memory in the brain. Toward this end, Lashley performed additional experiments in which he trained rats to navigate a maze and then destroyed different parts of their brains. He found that no matter where the brain damage was located, rats still retained some memory of the maze (Lashley, 1929, 1943, 1950). Lashley interpreted these results as evidence that it was the amount of cortical tissue removed, not its location, that determined the degree of impairment in learning and memory. He also hypothesized that memories were widely distributed throughout the brain and that therefore there was no particular area of the brain that was especially critical for memory formation.
Lashley proposed two principles derived from his research on the physical basis of learning and memory: 1) mass action (the amount of cortex destroyed determines degree of impairment in learning and memory) and 2) equipotentiality (any part of cortex within a functional area can take over the functions of any other part of that same functional area).
Lashley's conclusions had two primary impacts. First, his analysis "tended to discredit, or at least deemphasize, the role of the interior [subcortical] parts of the brain in learning and memory and, on the other, tended to localize the mechanisms of learning and memory within the confines of the cerebral cortex" (Thompson, 1974). Second, Lashley's principles of mass action and equipotentiality led to an anti-localization bias in the study of learning and memory, which discouraged attempts to find specific brain structures involved in these functions. However, consistent with current views, and contradicting Lashley's findings, Kaada, Rasmussen, and Kveim (1961) reported that lesions of the hippocampo-fornix system impaired maze performance. This was an early study that confirms what we know today, that a subcortical structure, the hippocampus, is critical to many forms of learning and memory.
Hebb's Rule, Cell Assemblies, and the Engram
After receiving his doctorate from Harvard in 1936, one of Lashley's graduate students, Donald Hebb, published The Organization of Behavior: A Neuropsychological Theory. In this influential book, Hebb proposed that the "engram" consisted of changes at synapses during learning and memory formation. A quote from the book (Hebb, 1949, p. 62) explains Hebb's central principle of synaptic change in learning and memory:
“When an axon of cell A is near enough to excite a cell B and repeatedly or persistently takes part in firing it, some growth process or metabolic change takes place in one or both cells such that A’s efficiency, as one of the cells firing B, is increased."
Here Hebb is proposing an increase in the strength of synaptic connections between neurons that consistently fire together and that this involves growth or metabolic events in one or both of the cells.
More simply stated: "Neurons that fire together, wire together." This is known as "Hebb's rule" (retrieved from https://can-acn.org/donald-olding-hebb/ August 8, 2021).
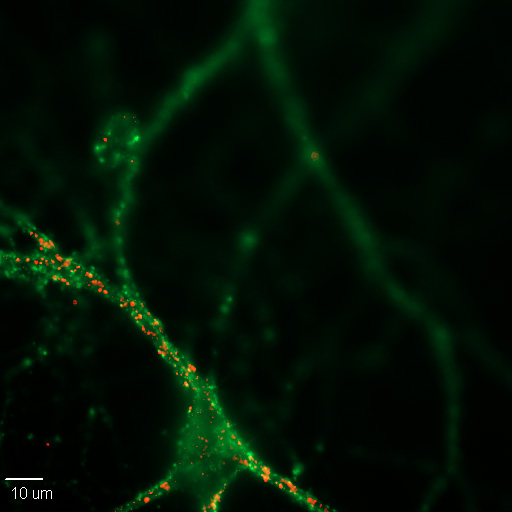
Figure \(\PageIndex{1}\): A 20X image of a cultured mouse cortical neuron in cell culture. Synapses are labeled for pre- (green) and post-synaptic (red) proteins, synaptophysin and PSD-95, respectively. (Image and caption from Wikimedia, Synapse; https://commons.wikimedia.org/wiki/F...n_Synapses.jpg; author, Dchordpdx; licensed under the Creative Commons Attribution 4.0 International license).
Neuroscientists use the term, Hebbian synapses, to refer to synapses that follow this principle. Hebb also proposed the idea of "cell assemblies." Hebb hypothesized that when cells repeatedly firing together, wired together, eventually they would form larger structures, "cell assemblies," which would form neural representations of whole, complex perceptions, ideas, memories, and other cognitive structures, such as schemas, and highly abstract categories and concepts. All of this could be built from changes in the efficiency of synaptic transmission at specific synapses, following Hebb's principle that "neurons that fire together, wire together." One can imagine that entire cell assemblies that fire together might also wire together, creating much larger and much more complex neural representations in the brain of complex, abstract ideas including such things as scientific theories and mathematical formulations.
Non-Association Learning in Aplysia
These ideas have inspired the hunt by neuroscientists for changes at synapses and in pre- or post-synaptic neurons, during and after learning--changes which might be the physical basis in the nervous system for learning and memory (Lashley's "engram," which, by the way, has nothing to do with the non-scientific use of the term by Scientology). Much of the pioneering scientific work on the search for the "engram" has involved looking for these physical changes associated with learning in the simple nervous system of a marine species, Aplysia californica, a type of sea slug. A major reason for the selection of this species for study, aside from the simplicity of its nervous system (just 20,000 neurons), is the large size of its neurons which makes the work and the observations easier.

Figure \(\PageIndex{2}\): Aplysia californica. (Image from Wikimedia, Aplysia californica; https://commons.wikimedia.org/wiki/F...,_Monterey.jpg; Chad King / NOAA MBNMS; this image is in the public domain because it contains materials that originally came from the U.S. National Oceanic and Atmospheric Administration, taken or made as part of an employee's official duties).
Eric Kandel (1976) and his colleagues did pioneering work which showed synaptic changes that mediate habituation (see sections above) of the Aplysia's siphon and gill defensive withdrawal reflex. A decrease in conductivity was found at synapses involved in the siphon and gill withdrawal reflex in Aplysia after habituation to a repeated presentation of a harmless novel stimulus. Kandel and co-workers showed that the pre-synaptic sensory neuron released less neurotransmitter onto the post-synaptic motor neuron as a result of repeated presentation of the novel stimulus leading to habituation of the reflex. The opposite was found for increased responsiveness. Sensitization refers to enhancement of responsiveness to a familiar stimulus. A single small electric shock to the tail of Aplysia heightens its gill withdrawal response for minutes to hours. Kandel and colleagues found that sensitization was mediated by an increase in release of neurotransmitter from the pre-synaptic sensory neuron onto the post-synaptic motor neuron serving the gill muscle. Thus, sensitization is the opposite of habituation both behaviorally and at the level of transmitter release (Kolb and Whishaw, 2001). Later studies showed synaptic changes involving modified transmitter release during classical conditioning as well (Kandel and Schwartz, 1982).
Hebb's Dual-Trace Theory of Memory
Hebb also proposed a dual-trace theory of memory--a short-term dynamic electrical process for short-term memory (Hebb referred to "reverberating circuits" holding this brief electrical activity) and, following sometime later, an enduring structural change at synapses as the physical basis (the engram) for long-term memory (Hebb, 1949). Decades of research followed using various agents, including electroconvulsive shock (ECS), shortly after learning in rats to disrupt the short-term memory trace, which according to Hebb, consisted of circulating or "reverberating" electrical activity, thus preventing long-term storage. However, although ECS in rats within about 30 seconds of learning seemed to disrupt consolidation of the short-term memory trace into a permanent long-term trace as evidenced by apparent amnesia for the learning task, additional research suggested alternative explanations for these results. One alternative was that the ECS did not prevent consolidation of the long-term memory trace, but instead disrupted retrieval of the memory. Evidence for this failure of retrieval hypothesis suggests that the original failure of consolidation interpretation of the ECS research was likely incorrect (Miller and Martin, 2014).
The Special Roles of the Hippocampus, Cerebellum, and Amygdala
As discussed above, research since Lashley has revealed that his speculation that all areas of brain were equally involved in learning and memory was incorrect. Now we know that one of the most important brain regions in explicit memory is the hippocampus, which serves as a preprocessor and elaborator of information (Squire, 1992). The hippocampus helps us encode information about spatial relationships, the context in which events were experienced, and the associations among memories (Eichenbaum, 1999). The hippocampus also serves in part as a switching point that holds the memory for a short time and then directs the information to other parts of the brain, such as the cortex, to actually do the rehearsing, elaboration, and long-term storage (Jonides, Lacey, & Nee, 2005; Saylor Foundation, 2015). We also now know that different parts of the brain are involved in different kinds of memory.
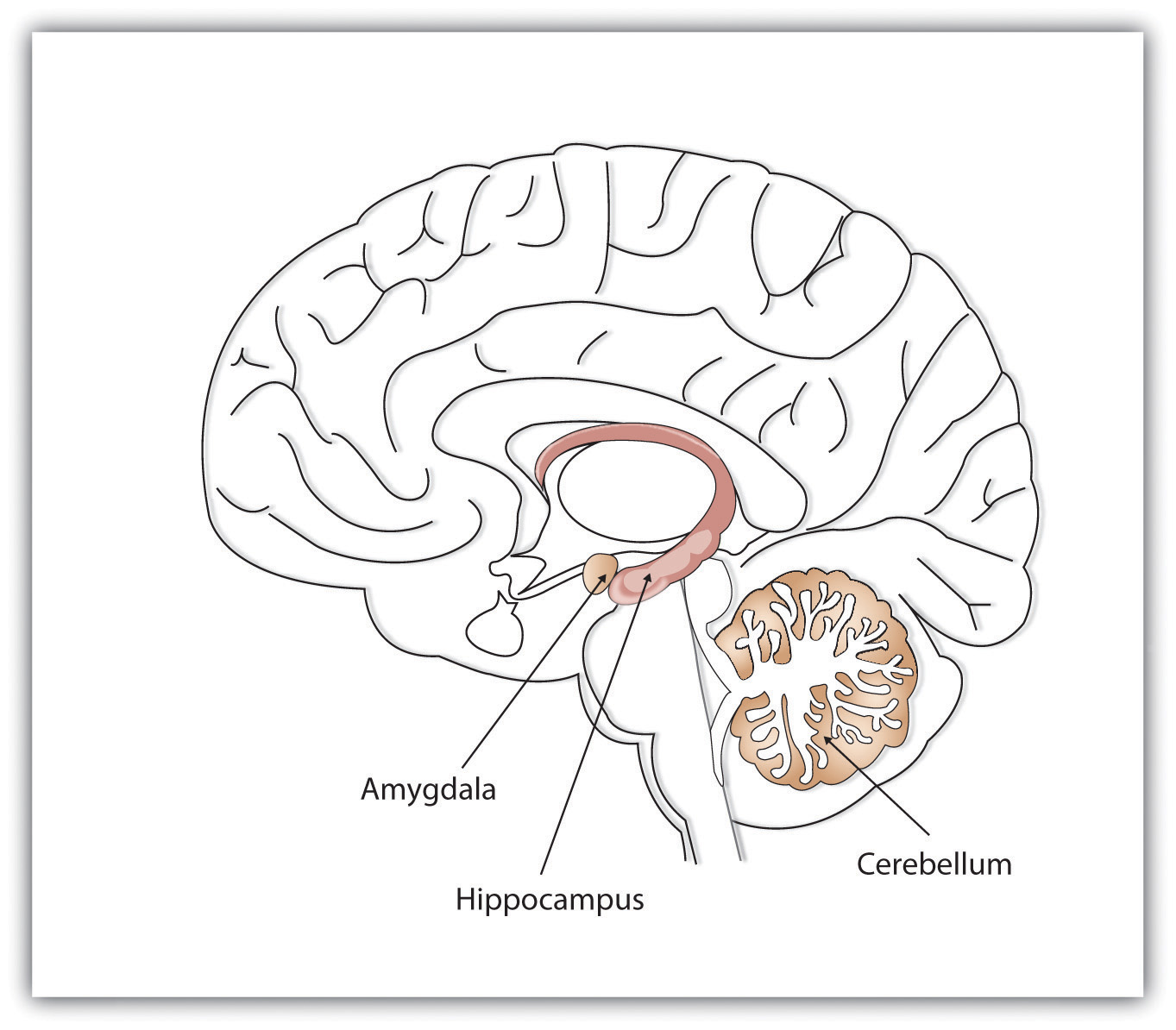
Figure \(\PageIndex{3}\): Different brain structures help us remember different types of information. The hippocampus is particularly important in explicit memories, the cerebellum in implicit memories, and the amygdala in emotional memories (Image and caption from the Saylor Foundation, 2015; Remembering and Judging; https://learn.umgc.edu/d2l/le/conten.../25917628/View; licensed under a Creative Commons Attribution 3.0 Unported License.).
While the hippocampus is handling explicit memory, the cerebellum and the amygdala are critically involved in implicit and emotional memories, respectively. Research shows that the cerebellum is more active when we are learning associations and in priming tasks (based on implicit memory), and animals and humans with damage to the cerebellum are impaired in classical conditioning (Krupa, Thompson, & Thompson, 1993; Woodruff-Pak, Goldenberg, Downey-Lamb, Boyko, & Lemieux, 2000). The storage of many of our most important emotional memories, and particularly those related to fear, is initiated and controlled by the amygdala (Sigurdsson, Doyère, Cain, & LeDoux, 2007).
Changes at the Synapse Correlated with Learning and Memory
Hebb's theory that long-term memory was stored by physical changes at the synapse has been so influential that research on the neural basis of learning and memory has focused primarily on synaptic events (see Chapter 5 on synapses and synaptic transmission). Focus on the synapse was reinforced by the discovery in 1973 in rabbit hippocampus that a long-lasting increase in synaptic conductivity (synaptic strength) could be produced by high frequency stimulation of the pre-synaptic neuron (Bliss and Lomo, 1973). This finding that synaptic strength can be increased for an extended period of time following high frequency pre-synaptic stimulation is now referred to as Long-Term Potentiation (LTP). LTP has been found in many species and in many parts of the brain. However, it has been studied most in the hippocampus of the rat. Since this early research, additional forms of synaptic change have also been discovered.
Synaptic Plasticity
Synaptic plasticity is the strengthening or weakening of synapses over time in response to increases or decreases in their activity. Plastic change also results from the alteration of the number of receptors located at a synapse. Synaptic plasticity is the basis of learning and memory, enabling a flexible, functioning nervous system. Synaptic plasticity can be either short-term (synaptic enhancement or synaptic depression) or long-term. Two processes in particular, long-term potentiation (LTP) and long-term depression (LTD), are important forms of synaptic plasticity that occur in synapses in the hippocampus.
Synaptic Plasticity: short-term enhancement, long-term potentiation and long-term depression
Key Points
- Short-term synaptic enhancement occurs when the amount of available neurotransmitter is increased, while short-term synaptic depression occurs when the amount of vesicles with neurotransmitters is decreased.
- Synapses are strengthened in long-term potentiation (LTP) when AMPA receptors (which bind to negatively-charged glutamate) are increased, allowing more calcium ions to enter the cell, causing a higher excitatory response.
- Long-term depression (LTD) occurs when the AMPA receptors are decreased, which decreases the amount of calcium ions entering the cell, weakening the synaptic response to the release of neurotransmitters.
- The strengthening and weakening of synapses over time controls learning and memory in the brain.
Key Terms
- long-term potentiation: a long-lasting (hours in vitro, weeks to months in vivo) increase, typically in amplitude, of the response of a postsynaptic neuron to a pattern of high frequency stimuli from a presynaptic neuron
- long-term depression: a long-term weakening of a synaptic connection
- plasticity: the property of neuron's conductivity or synaptic strength that allows it to be strengthened or weakened
- NMDA receptor: N-methyl-D-aspartate (NMDA) post-synaptic receptor and ion channel that is activated when Glutamate transmitter binds to it; an ionotropic (see Chapter 5) glutamate receptor
- AMPA receptor: Alpha-Amino-3-Hydroxy-5-Methyl-4-Isoxazole Propionic Acid (AMPA) post-synaptic receptor and ion channel that is activated when Glutamate transmitter binds to it; an ionotropic (see Chapter 5) glutamate receptor; both NMDA and AMPA receptors are important in learning and memory
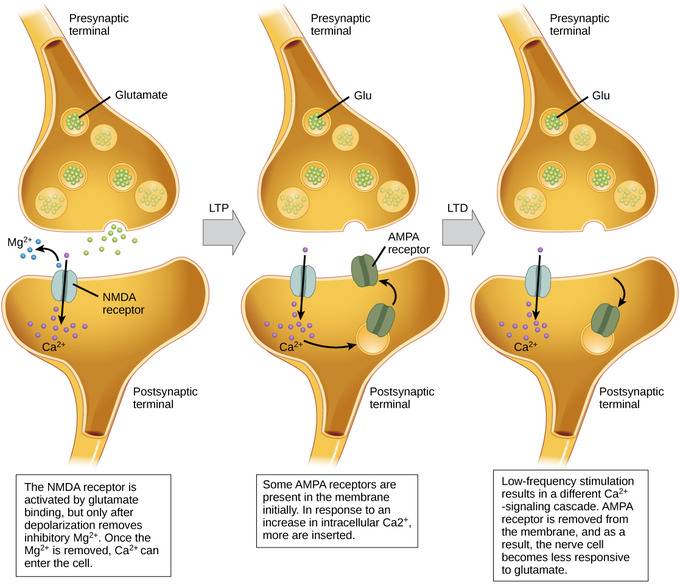
Figure \(\PageIndex{4}\): Long-term potentiation and depression: Calcium entry through postsynaptic NMDA receptors can initiate two different forms of synaptic plasticity: long-term potentiation (LTP) and long-term depression (LTD). LTP arises when a single synapse is repeatedly stimulated. This stimulation causes a calcium- and CaMKII-dependent cellular cascade, which results in the insertion of more AMPA receptors into the postsynaptic membrane. The next time glutamate is released from the presynaptic cell, it will bind to both NMDA and the newly-inserted AMPA receptors, thus depolarizing the membrane more efficiently. LTD occurs when few glutamate molecules bind to NMDA receptors at a synapse (due to a low firing rate of the presynaptic neuron). The calcium that does flow through NMDA receptors initiates a different calcineurin and protein phosphatase 1-dependent cascade, which results in the endocytosis of AMPA receptors. This makes the postsynaptic neuron less responsive to glutamate released from the presynaptic neuron. (Image and caption from Lumen Boundless Biology, How Neurons Communicate; https://courses.lumenlearning.com/bo...s-communicate/; curation and Revision. Provided by: Boundless.com. License: CC BY-SA: Attribution-ShareAlike.)
Long-term Potentiation (LTP)
Long-term potentiation (LTP) is a persistent strengthening of a synaptic connection, which can last for minutes or hours or even weeks. LTP is based on the Hebbian principle: “cells that fire together wire together.” There are various mechanisms, none of which are fully understood, behind the synaptic strengthening seen with LTP.
One known mechanism involves a type of postsynaptic glutamate receptor: NMDA (N-Methyl-D-aspartate) receptors. These receptors are normally blocked by magnesium ions. However, when the postsynaptic neuron is depolarized by multiple presynaptic inputs in quick succession (either from one neuron or multiple neurons), the magnesium ions are forced out and Ca2+ ions pass into the postsynaptic cell. Next, Ca2+ ions entering the cell initiate a signaling cascade that causes a different type of glutamate receptor, AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) receptors, to be inserted into the postsynaptic membrane. Activated AMPA receptors allow positive ions to enter the cell.
Therefore, the next time glutamate is released from the presynaptic membrane, it will have a larger excitatory effect (EPSP) on the postsynaptic cell because the binding of glutamate to these AMPA receptors will allow more positive ions into the cell. The insertion of additional AMPA receptors strengthens the synapse so that the postsynaptic neuron is more likely to fire in response to presynaptic neurotransmitter release.
LTP has many similarities with the synaptic changes Hebb proposed as basis for long-term memory, including two key features. First, LTP is long lasting, up to a year with repeated trials. Second, many forms of LTP require simultaneous activation in pre-synaptic and post-synaptic neurons at the synapse where LTP takes place ("neurons that fire together, wire together"); this is because NMDA (N-methyl-d-aspartate) receptors for glutamate (the most common excitatory transmitter in the brain), which are prominent at synapses where LTP occurs, have the same requirement for simultaneous activity in both pre- and post-synaptic neurons in order for these receptors to become activated. Research showing that many features of LTP are similar to features of long-term memory provides strong circumstantial evidence that LTP is related to the mechanisms of learning and memory. For example, LTP can be stimulated by low intensity stimulation similar to that produced in single neurons; LTP is most prominent in structures associated with learning; LTP is produced in the hippocampus by learning; drugs that enhance or impair learning also enhance or impair LTP; and LTP occurs in the nervous system of simple invertebrates (see discussion of Aplysia above) at the specific synapses involved in the learning (Pinel & Barnes, 2021).
Long-term Depression (LTD)
Another related phenomenon is long-term depression (LTD), associated with decreases in synaptic conductivity. LTD might be part of the processes involved in creating and modifying patterns of excitation and inhibition in large populations of neurons for the coding of learned movements, sensory experience, and perhaps the mental representations of complex cognitive structures such as perceptions, whole memories, concepts, and even abstract ideas (Churchland, 2013). Coding of mental events in terms of patterns of excitation and inhibition in large populations of neurons is discussed later in this chapter when we examine neural network modeling of learning and memory. A related possibility is the pruning away of some synapses to form permanent changes in neural circuitry.Long-term depression (LTD) is essentially the reverse of LTP: it is a long-term weakening of a synaptic connection. One mechanism known to cause LTD also involves AMPA receptors. In this situation, calcium that enters through NMDA receptors initiates a different signaling cascade, which results in the removal of AMPA receptors from the postsynaptic membrane. With the decrease in AMPA receptors in the membrane, the postsynaptic neuron is less responsive to the glutamate released from the presynaptic neuron. While it may seem counterintuitive, LTD may be just as important for learning and memory as LTP. The weakening and pruning of unused synapses trims unimportant connections, leaving only the salient connections strengthened by long-term potentiation.
Short-term Synaptic Enhancement and Depression
Short-term synaptic plasticity acts on a timescale of tens of milliseconds to a few minutes. Short-term synaptic enhancement results from more synaptic terminals releasing transmitters in response to presynaptic action potentials. Synapses will strengthen for a short time because of either an increase in size of the readily- releasable pool of packaged transmitter or an increase in the amount of packaged transmitter released in response to each action potential. Depletion of these readily-releasable vesicles causes synaptic fatigue. Short-term synaptic depression can also arise from post-synaptic processes and from feedback activation of presynaptic receptors.
Changes at the Synapse Correlated with Learning and Memory: Anatomical Changes in Dendritic Spines
Modifications in synaptic strengths in excitatory synapses in the hippocampus appear to play a critical role in storage and recall of information in mammals. In humans at least, the role of the hippocampus is especially critical in explicit episodic (autobiographical) memory. As noted above, it is also crucially involved in spatial memory for many mammal species, including humans. Changes in synaptic strength throughout much of the brain may be important in learning and memory.
Dendritic Spines
Dendritic spines are tiny protruding structures located on the shaft of dendrites and are associated with synaptic connections. These spines "are present in large numbers on the surface of dendrites. For example, a single pyramidal neuron in the hippocampal CA1 region possesses as many as 30,000 dendritic spines. A majority of excitatory synapses are formed on the surface of these dendritic spines" (Irie & Yamaguchi, 2009, p. 1141). The primary sites of excitatory synaptic interaction in the mammalian central nervous system appear to be at dendritic spines.
These spiny protrusions have a head and neck of varied morphology (see Figures 10.4.5 and 10.4.6). Changes in spines play a significant role during brain development. "In a developing brain, spines exhibit a high degree of structural and functional plasticity, reflecting the formation and elimination of synapses during the maturation of neuronal circuits. The morphology of spines in developing neurons is affected by synaptic activity, hence contributing to the experience-dependent refinement of neuronal circuits, learning, and memory. Thus, understanding spine dynamics and its regulation is of central importance to studies of synaptic plasticity in the brain" (Bertling, et al., 2012, p. 391). During brain development synapses are formed, modified and sometimes eliminated as a function of input to them.
As discussed above, LTP has been shown to lead to functional changes at synapses where it is induced. These changes due to LTP are increased synaptic conductivity and enhanced responsiveness of the post-synaptic neuron. It appears that anatomical changes in spines accompany these changes in synaptic strength which occur during LTP and learning and memory.
Remember Hebb's proposal that long-term memory storage involves some persistent anatomical change at synapses. If Hebb was right, then we should expect to see anatomical changes at synapses when learning and memory occur. And if LTP is involved in the formation of these hypothesized anatomical changes, then we should expect to see anatomical modifications at the synapse associated with the LTP-induced increases in synaptic conductivity and responsiveness of the post-synaptic neuron. Consistent with Hebb's hypothesis about the physical basis of learning and memory, this is just what neuroscientists have observed. Changes in synaptic strength, associated with LTP, are accompanied by LTP-induced alterations of the shape and size of dendritic spines (Chidambaram, et al., 2019; Harris, et al., 2003), anatomical changes at the synapse, just as Hebb predicted.
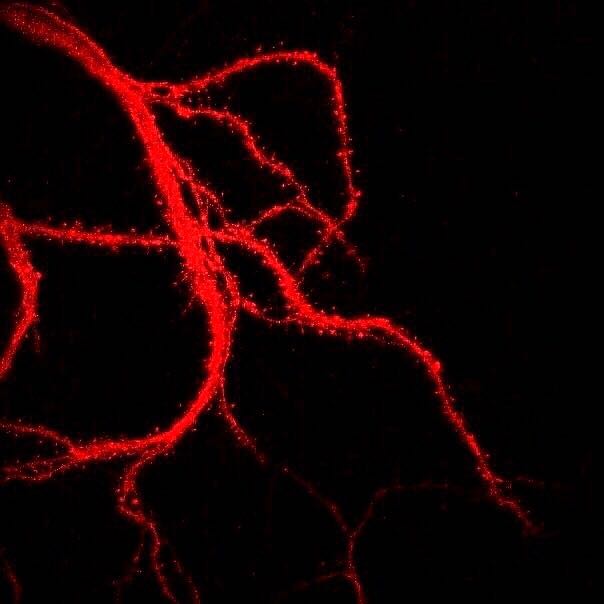
Figure \(\PageIndex{5}\): Branching dendrites of a neuron showing dendritic spines (tiny bristle-like projections lining each dendritic branch). The photograph was obtained with a laser scanning microscope. Dendritic spines can rapidly change in size and shape and numbers, and are important in learning and memory. (Caption by Kenneth A. Koenigshofer, PhD. Image from Wikimedia Commons; File:Нейрональные отростки с шипиками.jpg; https://commons.wikimedia.org/wiki/F...0%BC%D0%B8.jpg; by Sergb95; licensed under the Creative Commons Attribution-Share Alike 4.0 International license).
Evidence strongly suggests that new protein synthesis is necessary for these effects on the shape and size of dendritic spines (Chidambaram, et al., 2019). As noted above, both growth and metabolic changes at synapses were proposed by Hebb as possible basis for the long-term memory. These anatomical changes in dendritic spines induced by LTP can be long-lasting. For example, "In mature networks, synaptic connections at dendritic spines can be quite stable, as newly emergent spines generated after motor learning have been shown to persist for months. . . While the distribution of spine size across the dendritic arbor of a single neuron can be quite variable, spine size generally correlates with excitatory synapse strength . . . it is generally accepted that spine head diameter and synapse strength co-vary during the expression of long term potentiation (LTP) . . ." (Henry, et al., 2017). It appears that Hebb's hypothesis about the physical basis of learning and memory in the brain was quite predictive of what later research would show--one mark of a good scientific theory.
Consistent with Hebb's hypothesis, dendritic spines appear to function as storage sites for synaptic strength and help transmit electrical potentials to the neuron's cell body. Dendrites of a single neuron can contain hundreds, even thousands of spines. Dendritic spines usually receive excitatory input from axons, although sometimes both inhibitory and excitatory connections are made onto the same dendritic spine (Kasthuri, et al., 2015). In addition to spines providing a potential anatomical substrate for memory storage and synaptic transmission, they may also increase the number of possible contacts between neurons (Alvarez & Sabatini, 2007). Spines are found on the dendrites of most principal neurons in the brain, including the pyramidal neurons of the neocortex (in prefrontal cortex for cognition; corticospinal tract for movement), the medium spiny neurons (GABAergic inhibitory) of the striatum (striate nucleus of basal ganglia; motor and reward systems; receives Glutamatergic and Dopaminergic inputs), and the Purkinje cells (GABAergic inhibitory neurons) of the cerebellum which receive relatively weaker excitatory (Glutamatergic) synapses to spines on the Purkinje cell dendrite. Hippocampal and cortical pyramidal neurons may receive tens of thousands of mostly excitatory inputs from other neurons onto their spines, whereas the number of spines on Purkinje neuron dendrites in the cerebellum is even greater, up to ten times greater.
Three of the four most notable classes of spine shape are shown in the figure below: "thin", "stubby", and "mushroom." The fourth category, "branched", is not shown. Studies using electron microscopy have revealed a continuum of shapes between these categories (Ofer, et al., 2021). The variable spine shape and volume is correlated with the strength and maturity of each spine-synapse. "Dendritic spines are also highly motile undergoing changes in size and shape over a timescale of seconds to minutes. Because small, thin dendritic spines are most likely to undergo these structural changes, whereas large, so-called ‘mushroom’ spines tend to maintain their form, it has been suggested that mushroom spines are more stable memory spines whereas the more plastic thin spines are learning spines. . . . [L]arge spines are sites of strong synapses and, accordingly, the growth of the spine head likely correlates with a strengthening of synaptic transmission" (Leuner & Shors, 2010, p. 349).
Both shorter-term transient and longer-term sustained changes in structural plasticity of dendritic spines have been observed by researchers--within the first two minutes following stimulation of the pre-synaptic neuron, an initial 300% expansion of the post-synaptic dendritic spine occurs, followed by a reduction of the spine's volume so that it is elevated to about 70-80% larger than the original pre-stimulation volume, a sustained change in structural plasticity lasting about 30 minutes or more. LTP is initiated with the transient stage of dendritic spine growth. Both stages of spine growth are hypothesized to be involved in learning and memory. Large spines are more stable than smaller ones and may be resistant to modification by additional synaptic activity (Kasai, et al., 2003). These structural changes in dendritic spines induced by pre-synaptic stimulation associated with LTP are believed by neurosciences to be important in the encoding, storage, and retrieval of memories (Murakoshi, et al., 2011). These changes in spine size and shape involve changes in the microstructure of the spine and abnormalities at this level may be involved in memory disorders. "The actin cytoskeleton is the structural element underlying changes in dendritic spine morphology and synapse strength. The proper morphology of spines and proper regulation of the actin cytoskeleton have been shown to be important in memory and learning; defects in regulation lead to various memory disorders. Thus, understanding actin cytoskeleton regulation in dendritic spines is of central importance to studies of synaptic and neuronal function" (Koskinen, et al., 2012, p. 47).
The morphogenesis of dendritic spines (the process by which they attain their shape) is critical to the induction of long-term potentiation (LTP) (Kim & Lisman, 1999; Krucker, et al., 2000). Interestingly, Hayashi-Takagi et al. (2015) found that memory could be disrupted if the potentiated spines, within a neuron ensemble involved in motor learning, were specifically shrunk. This suggests that spine growth is essential to at least some forms of memory.

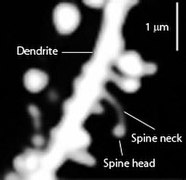
Figure \(\PageIndex{6}\): (Left) Varying morphology of types of dendritic spines. Changes in their shape and size are associated with LTP and learning and memory. (Right) Spines on the dendrite of a medium spiny striatal neuron. The image was obtained by expressing Enhanced Green Fluorescent Protein (EGFP) in the neurons and imaging them using a laser scanning two photon microscope. (Caption for image on left by Kenneth A. Koenigshofer, PhD; Image on left from Wikimedia Commons, Dendritic spines; https://commons.wikimedia.org/wiki/F...e_types_3D.png; original work of Thomas Splettstoesser (www.scistyle.com); licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license. Image on right and its caption from Wikimedia Commons, Dendritic spines; https://commons.wikimedia.org/w/inde...=Go&type=image; Released into Public Domain by the image author).
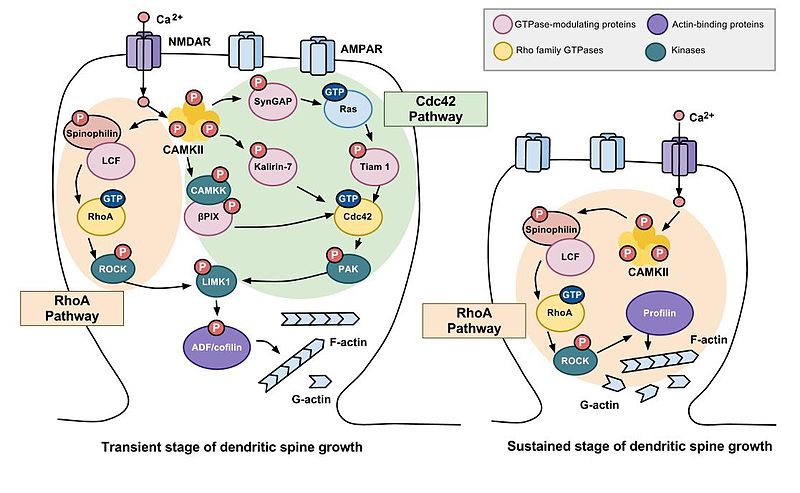
Figure \(\PageIndex{7}\): Transient vs. Sustained Dendritic Spine Growth following High-Frequency Stimulation. Details inside each spine show the chemical cascades involved in the two stages of spine growth after pre-synaptic stimulation. It is not necessary for the student to know the details of these cascades. However, note that these cascades begin with calcium influx through NMDA receptors (top of spine) and that the final step is action on actin (bottom right of spine) leading to changes in shape and size of the spine. (Image from Wikimedia Commons; Dendritic spines; https://commons.wikimedia.org/wiki/F...timulation.jpg; author of image and image title, Itzy; licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license; caption by Kenneth A. Koenigshofer, PhD.).
Yang, et al. (2009) studied changes in numbers of synaptic spines during learning and novel sensory experience in mice. They state that their "results suggest that new experience leads to the pruning of existing synapses and could cause significant functional changes in cortical circuits. Indeed, we found that 1 week after motor training, motor performance strongly correlated with the degree of spine elimination . . . Thus, motor learning and novel sensory experience involve not only new spine formation but also permanent removal of connections established early in life" (Yang, et al. 2009, p. 921). These results are easier to understand if we recall that during prenatal development of the brain there are excess numbers of synaptic connections and that the elimination of nonfunctional synapses is essential to normal brain development and functioning. Learning may involve neural pruning as well as the formation and enhanced conductivity of synapses via changes in dendritic spines. As Yang, et al., note, "In addition to promoting synapse formation, experience plays an important role in eliminating excessive and imprecise synaptic connections formed early during development" (Yang, et al. 2009, p. 921).
Chidambaram, et al. (2019, p.161) summarize the relationship of changes in synaptic conductivity to changes in dendritic spines: "During synaptic plasticity the number and shapes of dendritic spines undergo radical reorganizations. Long-term potentiation (LTP) induction promotes spine head enlargement and the formation and stabilization of new spines. Long-term depression (LTD) results in their shrinkage and retraction." These authors also note some interesting observations relating atypical numbers of synaptic spines to several neurologically based disorders. "Reports indicate increased spine density in the pyramidal neurons of autism and Fragile X syndrome patients and reduced density in the temporal gyrus loci of schizophrenic patients. Post-mortem reports of Alzheimer's brains showed reduced spine number in the hippocampus and cortex" and atypical spines may play a role in neurodegenerative diseases (Chidambaram, et al., 2019, p. 161).
If changes in spines are involved in the formation and retention of long-term memories, then morphological changes in spines must be very durable. According to Yang and colleagues (2009, p. 920), "Stably maintained dendritic spines are associated with lifelong memories. . . . a small fraction of new spines induced by novel experience, together with most spines formed early during development and surviving experience-dependent elimination, are preserved throughout the entire life of an animal. These studies indicate that learning and daily sensory experience leave minute but permanent marks on cortical connections and suggest that lifelong memories are stored in largely stably connected synaptic networks."
Hebb's theory has proven to be impressively predictive of discoveries made many years after his book was published in 1949. Findings like those described in this module continue to confirm many aspects of Hebb's theory.
Cellular and Receptor Level Mechanisms Revisited
In the sections above, we discussed Long-Term Potentiation (LTP) and its possible involvement in memory processes. Here, it is appropriate to consider LTP and associated processes in greater detail. As noted previously, Long-term potentiation (LTP) is a process in which synapses are strengthened. It has been the subject of extensive research since the mid-1970s because of its likely role in several types of memory.
A related phenomenon also discussed above is Long-Term Depression (LTD), a phenomenon that is the opposite of long-term potentiation (LTP). In LTD, communication across the synapse is silenced. LTD plays an important role in the cerebellum, in implicit procedural memory (motor memory, implicit memory of how to do something like ride a bicycle or hit a baseball), where the neural networks involved in erroneous movements are inhibited by the silencing of their synaptic connections. LTD is what allows us to correct our motor procedures when learning how to perform a task. Thus, long-term depression (LTD) involves a weakening of a synapse as a means of improving performance of learned skilled movements (recall that weakening of synaptic strength is associated with shrinkage of dendritic spines). Imagine that you are trying to learn a gymnastics routine that involves a number of flips and other complicated movements in sequence. When you initially start to learn the routine, you make many incorrect moves that you must suppress if you are going to learn to do the routine correctly with the error free precision that the judges will look for. LTD is the cellular level mechanism that allows you to suppress the incorrect muscle movements that lead to errors in the routine.
As discussed previously, there is another possible role for LTD in coding of perceptions, concepts, ideas, and other complex cognitive structures. One of the most difficult problems in neuroscience is how the activities of large populations of neurons encode and create complex elements of psychological experience such as perceptions, thoughts, ideas, understanding, knowledge, and other complex mental representations. Modeling of brain processes in artifical neural networks (see the supplement later in this chapter on artificial networks) suggests that inhibition is just as important as excitation in neural coding of complex mental representations. Patterns of excitation and inhibition in large populations of neurons may underlie the neural coding of complex cognition and perception. If so, then LTD may be equally as important as LTP in the neural coding of complex representations such as perceptions, thoughts, concepts, ideas and even emotions (see Matter and Consciousness by Paul Churchland for a more complete discussion of these ideas). Just as dots and dashes are equally important in Morse Code, excitation and inhibition may be equally important elements in neural coding.
Although a synapse is weakened in LTD , by contrast, in LTP, after intense stimulation of the pre-synaptic neuron, a synapse is strengthened and the amplitude of the post-synaptic neuron’s response increases. The stimulus applied to the pre-synaptic neuron to produce LTP is generally of short duration (less than 1 second) but high frequency (over 100 Hz). The excitatory potential (EPSP; see Chapter 5) measured in a post-synaptic neuron, after high frequency pre-synaptic stimulation, is increased for a long period. For example, when the axons that make connections to the pyramidal neurons of the hippocampus are exposed to a high-frequency stimulus, the amplitude of the excitatory potential measured in these pyramidal neurons is increased for up to several weeks. What synaptic events are involved in the production of LTP? Let's review, now in greater detail.
Glutamate, the neurotransmitter released into these hippocampal synapses (glutamate is excitatory and mediates fast synaptic neurotransmission in the brain), binds to several different sub-types of receptors on the post-synaptic hippocampal neuron. Two of these glutamate receptor sub-types, the receptors for AMPA and NMDA, are especially important for LTP (Traynelis, et al., 2010).
The AMPA receptor (a glutamate receptor named after the derivative that activates it, AMPA short for Alpha-Amino-3-Hydroxy-5-Methyl-4-Isoxazole Propionic Acid) is paired with an ion channel so that when glutamate binds to this receptor, this channel lets sodium ions enter the post-synaptic neuron. This influx of sodium causes the post-synaptic dendrite to become locally depolarized (an EPSP, a positive shift in voltage).
The NMDA receptor, which also uses glutamate as its transmitter, is also paired with an ion channel (NMDA is N-methyl-D-aspartate, a derivative of glutamate). This channel admits calcium ions into the post-synaptic cell when it is activated. However, when the cell is at resting potential, the calcium channel is blocked by magnesium ions (Mg2+), so that even if glutamate binds to the receptor, calcium cannot enter the neuron. For these magnesium ions to withdraw from the channel, the dendrite’s membrane potential must be depolarized. And that is exactly what happens during the high-frequency stimulation that causes LTP: the post-synaptic neuron becomes depolarized following the sustained activation of its AMPA receptors! The magnesium then withdraws from the NMDA receptors and allows large numbers of calcium ions to enter the cell.
This increased concentration of calcium in the dendrite sets off several biochemical reactions that make this synapse more efficient for an extended period (Bliss & Collingridge, 1993; Bliss, et al., 2018; Citri & Malenka, 2008). These calcium ions are extremely important intracellular messengers that activate many enzymes by altering their conformation. One of these enzymes is calmoduline, which becomes active when four calcium ions bind to it. It then becomes Ca2+/calmodulin, the main second messenger for LTP. Ca2+/calmodulin then in turn activates other enzymes that play key roles in this process, such as adenylate cyclase and Ca2+/calmodulin-dependent protein kinase II (CaM kinase II). These enzymes in turn modify the spatial conformation of other molecules, usually by adding a phosphate ion to them. This common catalytic process is called phosphorylation.
The activated adenylate cyclase manufactures cyclic adenosine mono-phosphate (cAMP), which in turn catalyzes the activity of another protein, kinase A (PKA). In short, there is a typical cascade of biochemical reactions which can have many different effects.
For example, PKA phosphorylates the AMPA receptors, allowing them to remain open longer after glutamate binds to them. As a result, the post-synaptic neuron becomes further depolarized, thus contributing to LTP.
Other experiments have shown that CREB protein is another target of PKA, protein kinase A. CREB plays a major role in gene transcription, and its activation leads to the creation of new AMPA receptors that can increase synaptic efficiency still further.
According to Park, et al. (2021), Long-term potentiation (LTP) at hippocampal CA1 synapses (see Figure 10.4.8) can be expressed by an increase either in the number (N) of AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid) receptors or in their single channel conductance (γ)--that is, increased synaptic strength. In their experiments, they established how these distinct synaptic processes contribute to the expression of LTP in hippocampal slices obtained from young adult rodents. LTP induced by compressed theta burst stimulation (TBS), with a 10 second inter-episode interval, involves purely an increase in the number of AMPA receptors (LTPN). In contrast, either a spaced TBS, with a 10 min inter-episode interval, or a single TBS, delivered when PKA is activated, results in LTP that is associated with a transient increase in single channel conductance (γ) or increased synaptic strength (LTPγ), caused by the insertion of calcium-permeable (CP)-AMPA receptors. Activation of CaMKII is necessary and sufficient for LTPN (increase in numbers of AMPA receptors, while PKA is additionally required for LTPγ (increase in single channel conductance). Thus, two mechanistically distinct forms of LTP co-exist at these synapses.
The other enzyme activated by Ca2+/calmodulin, CaM kinase II, has a property that is decisive for the persistence of LTP: it can phosphorylate itself! Its enzymatic activity continues long after the calcium has been evacuated from the cell and the Ca2+/calmodulin has been deactivated.
CaM kinase II can then in turn phosphorylate the AMPA receptors and probably other proteins such as MAP kinases, which are involved in the building of dendrites, or the NMDA receptors themselves, whose calcium conductance would be increased by this phosphorylation.
| LTP involves at least two phases: establishment (or induction), which lasts about an hour, and maintenance (or expression), which may persist for several days. | The first phase, establishment, can be experimentally induced by a single, high-frequency stimulation. It involves the activity of various enzymes (kinases) that persist after the calcium is eliminated, but no protein synthesis occurs. | To trigger the maintenance phase, however, a series of high-frequency stimuli must be applied. Unlike the establishment phase of LTP, the maintenance phase requires the synthesis of new proteins–for example, the ones that form the receptors and the ones that contribute to the growth of new synapses (another phenomenon that occurs during the maintenance phase of LTP). |
Table 10.4.1: Establishment and Maintenance Phases of LTP
To let the calcium enter the cell, the NMDA receptor must be activated by glutamate and subjected to depolarization simultaneously. The necessity for these two simultaneous conditions gives this receptor associative properties. This lets it detect the coincidence of two events and makes it the key element in long-term potentiation.
Significantly, if this receptor is blocked with a drug, or if the gene involved in its construction is disabled, LTP cannot occur.
The spines on post-synaptic dendrites form separate compartments to isolate biochemical reactions that occur at some synapses but not at others. This anatomical specialization probably helps to ensure a certain specificity in neural connections.
The most interesting characteristic of LTP is that it can cause the long-term strengthening of the synapses between two neurons that are activated simultaneously. In other words, exactly the kind of association mechanism that Hebb (1949) had imagined years earlier.
Experimental Evidence for Components of LTP
Many methods have been used to determine the role of a particular ion, or a second messenger, or an enzyme in a synaptic process.
For example, the role of calcium in long-term potentiation (LTP) has been confirmed in a number of ways. One experiment involved injecting the post-synaptic neuron with chelating agents such as EGTA and BAPTA, two molecules that bind to calcium and render it inactive. As a result, it becomes impossible to induce LTP. The reverse procedure has also been used. Researchers have injected special molecules into the post-synaptic neuron and then illuminated them with UV light, thus causing them to release enough calcium to induce LTP in this neuron.
Another approach is to produce mutations that make a protein non-functional or that block its action with another molecule. Blocking CaM kinase II in this way prevents LTP from becoming established, which also demonstrates the central role of this protein.
Similarly, inhibiting PKA or CREB prevents LTP from reaching its second phase and being sustained.
In certain cases, researchers have even identified the one amino acid, among the hundreds of amino acids that make up a protein, whose phosphorylation is essential for LTP (in case you want to know: Ser831 for the GluR1 sub-unit of the AMPA receptor and Thr286 for the autophosphorylation site of CaM kinase II).
Researchers have also shown that a mouse whose gene for the GluR1 sub-unit of the AMPA receptor had been knocked out could not have any LTP, thus confirming the role of CaM kinase II once again.
In mice for which the site Thr286 in CaM kinase II was deactivated, their basic synaptic transmission was maintained, but LTP could no longer be induced in them, thus proving the need for CaM kinase II. The reverse procedure also led to the same conclusion: adding activated CaM kinase II to the pyramidal neurons of the hippocampus causes a potentiation phenomenon similar to LTP.
Additional Mechanisms of LTP
“Silent synapses” are another mechanism that was discovered in the mid-1990s and that may contribute to long-term potentiation (LTP). These synapses are physically present, but under normal conditions do not contribute to synaptic transmission.
Some of these silent synapses have been found in the hippocampus. They appear to have receptors for NMDA but not for AMPA. It is thought that these synapses may be activated during LTP and thus help to strengthen the synaptic response. The discovery that after LTP, these synapses do display an electrical current associated with AMPA channels suggests that some newly synthesized AMPA receptors may be inserted into the post-synaptic membrane.
In addition to all of the post-synaptic mechanisms involved in the establishment of LTP, it has long been postulated that some pre-synaptic modifications occur during the ensuing maintenance phase. But certain modifications, such as an increase in the amount of glutamate released by the pre-synaptic neuron, would imply the presence of a retrograde messenger that goes back to this neuron and modifies it. Because nitric oxide (NO) is a gas in its natural state, and can thus diffuse through cell membranes, it would be an ideal candidate for this role. But its involvement is still the subject of much debate and controversy.
Video Reviews of LTP and Memory
For a review, take a look at these brief videos of the events in LTP:
https://www.youtube.com/watch?v=KyQUBukwwO8
LTP, Neurochemical Cascades, and Stages of Memory
According to Rozensweig (2007), different parts of the neurochemical cascade, associated with learning and memory, can be related to different stages in memory processing. Bennett, et al. (1964), made an early discovery that enriched experience in rats causes increased rates of protein synthesis and increased amounts of protein in the cortex. Mizumori, et al. (1985), using the protein-synthesis inhibitor, anisomycin, found that protein must be synthesized in the cortex soon after training if LTM is to be formed; however, short-term memory (STM) did not require protein synthesis, findings consistent with the two kinds of memory traces that Hebb (1949) had proposed: transient, labile memory traces on the one hand and stable structural traces on the other. Using chicks, several investigators traced a cascade of neurochemical events from initial sensory stimulation to synthesis of protein and structural changes in the brain (Rose, 1992). Rozensweig (2007) summarizes some of these events as follows:
"The cascade is initiated when sensory stimulation activates receptor organs that stimulate afferent neurons by using various synaptic transmitter agents such as acetylcholine (ACh) and glutamate. Inhibitors of ACh synaptic activity such as scopolamine and pirenzepine can prevent STM as can inhibitors of glutamate receptors including both the NMDA and AMPA receptors. Alteration of regulation of ion channels in the neuronal membrane can inhibit STM formation, as seen in effects of lanthanum chloride on calcium channels and of ouabain on sodium and potassium channels. Inhibition of second messengers is also amnestic, for example, inhibition of adenylate cyclase by forskolin or of diacylglycerol by bradykinin. These second messengers can activate protein kinases — enzymes that catalyze additions of phosphate molecules to proteins. We found that two kinds of protein kinases are important in formation, respectively, of ITM (an intermediate stage in memory formation noted by Rozenzweig, 2007) or LTM. Agents that inhibit calcium/calmodulin protein kinases (CaM kinases) prevent formation of ITM, whereas agents that do not inhibit CaM kinases, but do inhibit protein kinase A (PKA) or protein kinase C (PKC) prevent formation of LTM (Rosenzweig, et al., 1992; Serrano P.A., et al., 1994)."
Rose (1995) suggested that in chicks a kind of LTM that lasts a few hours (Rozensweig's ITM) involves a first wave of glycoprotein synthesis, whereas “true long-term memory” (LTM) requires a second wave of glycoprotein synthesis, occurring about 6 hours after training.
Rozensweig (2007) reviews evidence that the neurochemical cascades in memory in the chick are similar to the cascades in formation of LTP in different species. He states:
"The neurochemical cascade involved in formation of memory in the chick was soon shown to be similar to the cascade involved in long-term potentiation in the mammalian brain (Colley & Routtenberg, 1993) and in the nervous systems of invertebrates (Krasne & Glanzman, 1995). DeZazzo and Tully (1995) compared STM, ITM, and LTM in fruit flies, chicks, and rats. Tully and coworkers have shown that the three stages of memory in the fruit fly depend on three different genes (Tully et al., 1996)."
More recent research on LTP also confirms a three-stage process in LTP. In a review of the literature on LTP, Bliss, et al. (2018) note:
"The labels LTP1 and LTP2 equate to the forms of LTP that are, respectively, independent of and dependent on de novo protein synthesis. These are frequently referred to as early-phase LTP and late-phase LTP (E-LTP and L-LTP, respectively) implying that protein synthesis is not required initially but is required at later stages, with the switch-over [between E-LTP and L-LTP] occurring during a period of a few hours."
Although LTP has many properties that make it a good candidate for the mechanism of learning and memory in the brain, critical evidence in behaving animals is still needed. Nevertheless, neuroscientists are optimistic, and for good reason. According to Bliss, et al. (2018), "Today, LTP can be studied at every level from the purely molecular to the cognitive. Although definitive proof that the mechanisms of LTP subserve learning and memory in the behaving animal is still lacking, few neuroscientists doubt that such proof will eventually be forthcoming . . . there is now very strong evidence that an LTP-like mechanism mediates at least some aspects of memory." Much of this optimism comes from numerous studies showing that physiological, genetic, or pharmacological manipulations of LTP (either facilitating it or inhibiting it) have similar effects on learning and memory (Bliss, et al., 2018; Rozenzweig, 2007).
Anatomical Subregions of the Hippocampus and Memory
As mentioned above, hippocampal and cortical pyramidal neurons may receive tens of thousands of mostly excitatory inputs from other neurons onto their dendritic spines. As shown below in Figure \(\PageIndex{8}\), the hippocampus consists of a number of subregions. These regions are involved in different functions related to learning and memory. "The hippocampus proper is defined by the dentate gyrus and Cornu Ammonis (CA). While the dentate gyrus contains the fascia dentata and the hilus, the CA is anatomically and functionally differentiated into distinct subfields named CA1, CA2, CA3, and CA4. . . . [T]he CA3 subfield . . . with inputs from the dentate gyrus and entorhinal cortex . . . is implicated in encoding spatial representations and episodic memories. . . The mossy fiber pathway . . . translates . . . cortical signals to a . . . hippocampal code, essential for memory formation" (Cherubini & Miles, 2015, p.19). According to Yang et al. (2014), "CA3 pyramidal neurons form extensive recurrent connections with each other. Such connections are able to learn to associate components of an input pattern with each other." Perhaps this capacity would be required to link the components of a memory into a unified whole.
A prominent model of the organization of the hippocampus highlights what neuroscientists have dubbed the "trisynaptic circuits," circuits with three synapses (dentate--CA3--CA1), which according to the "lamellar hypothesis," are stacked upon one another along the body of the hippocampus. According to this model, "the hippocampus is organized as a stack of parallel, trisynaptic circuits" (Yang et al., 2014, p. 12919).
However, additional research suggests a network oriented perpendicular to the trisynaptic circuits. Yang et al. (2014, 12919-20) found a "well-organized, longitudinally projecting synaptic network among CA1 pyramidal neurons . . . [and] that synapses of this network are capable of supporting synaptic plasticity, including long-term potentiation, and a short-term memory mechanism called 'dendritic hold and read.' . . . [Furthermore,] LTP can strengthen interlamellar CA1-to-CA1 connections as well as the well-established CA3-to-CA1 connections in the transverse plane" (Yang et al., 2014, p. 12921). Might these two networks in planes perpendicular to one another in the hippocampus suggest a kind of grid-like arrangement which might code location of objects, including oneself, in three-dimensional space? As mentioned above, CA3 is implicated in the representation of space and spatial relationships. In addition, neuroscientists have found "place cells" in the hippocampus which fire when an animal is in a specific place in a maze, for example, and "place cells" tuned to fire to specific locations also seem to be present in the human hippocampus. According to Yang et al. (2014, p. 12923), "These properties suggest that this system may be an integral component of the larger 3D information processing network of the hippocampus." Clearly, processing of three-dimensional space and one's spatial relations to other objects is critical to navigating the world successfully.
However, Yang and colleagues consider another possibility--that the CA1 network may "transform a time sequence into a spatial sequence" so that the "resulting 'time-to-space' transform is effectively an efficient sequence memory mechanism" such as would be necessary when "information typically arrives as a time sequence over a finite interval and its meaning can only be revealed if each sequence is viewed as a whole and in proper order. . . [Furthermore,] behavioral studies have suggested that area CA1 possesses the capacity for sequence memory" (Yang et al., 2014, p. 12923). To understand what this means, think about events in the world--they take place over time and their meaning, including often their adaptive significance, may only become apparent when they are perceived as a whole sequence. If you introspect for a moment and consider what a memory is like when you recall it, you may get a sense of this. For example, I have a very distinct memory of the first time I saw the planet Saturn and its rings through a telescope I bought when I was 14 (which I still have) using money I had saved from delivering newspapers door-to-door on my paper route (yes, I am that old). After what is now 61 years ago, I still remember setting up my telescope on a cold night in the driveway of my parents' house, finding Saturn almost by accident, viewing the rings around the planet, and then running inside through the kitchen door to tell my parents what I was seeing. Could it be that the CA1 subregion of the hippocampus is an important component in the ability to remember events in their proper sequence? Sequence memory appears to be an essential property of the brain in order to represent and store vital information about the temporal order of events we experience in the world.
Figure \(\PageIndex{8}\): Diagram showing anatomy of the hippocampus. Pyramidal neurons in area CA1 have as many as 30,000 dendritic spines per neuron on their dendrites. Spines are associated with synapses which are primarily excitatory. The hippocampal subregion CA3-CA4 is indicated in black, stippled, and hatched areas. Black areas: suprapyramidal (SP), intra- and infrapyramidal (IIP) and hilar (CA4) mossy fiber terminal fields originating from the dentate gyrus. Stippled area: strata oriens (OR) and radiatum (RD). Hatched area: stratum lacunosum-moleculare (LM). CA1, subregion of the hippocampus without mossy fibers; FI, fimbria hippocampi; FD, fascia dentata; OL and ML, outer and middle molecular layers of the fascia dentata; SG, supragranular layer; GC, granular cells (Image and caption from Wikimedia Commons; File:Diagram of a Timm-stained cross-section of the hippocampus.JPEG; https://commons.wikimedia.org/wiki/F...ppocampus.JPEG; by Sluyter, Frans; Laure Jamot, Jean-Yves Bertholet, Wim Crusio (2005-04-22). "Prenatal exposure to alcohol does not affect radial maze learning and hippocampal mossy fiber sizes in three inbred strains of mouse". Behavioral and Brain Functions 1 (1): 5. DOI:10.1186/1744-9081-1-5. ISSN 1744-9081. Retrieved on 2007-12-21; licensed under the Creative Commons Attribution 2.0 Generic license).
References
Bennett E.L., et al. (1964). Chemical and anatomical plasticity of the brain. Science, 146:610. [PubMed]
Bertling, E., Ludwig, A., Koskinen, M., & Hotulainen, P. (2012). Methods for three-dimensional analysis of dendritic spine dynamics. In Methods in enzymology (Vol. 506, pp. 391-406). Academic Press.
Bliss T.V. & Collingridge G.L. (1993). A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 361 (6407): 31–39
Bliss, T. V., Collingridge, G. L., Morris, R. G., & Reymann, K. G. (2018). Long-term potentiation in the hippocampus: discovery, mechanisms and function. Neuroforum, 24 (3), A103-A120.
Cherubini, E., & Miles, R. M. (2015). The CA3 region of the hippocampus: how is it? What is it for? How does it do it?. Frontiers in cellular neuroscience, 9, 19.
Citri A., & Malenka R.C. (2008). Synaptic plasticity: multiple forms, functions, and mechanisms. Neuropsychopharmacology. 33 (1): 18–41.
Colley P.A., Routtenberg A. (1993). Long-term potentiation as synaptic dialogue. Brain Res Brain Res Rev. 18:115. [PubMed]
DeZazzo J., Tully T. (1995). Dissection of memory formation: from behavioral pharmacology to molecular genetics. Trends Neurosci. 18:212. [PubMed]
Hebb, D.O. (1949). The Organization of Behavior. New York. Wiley.
Irie, F., & Yamaguchi, Y. (2009). Eph receptor signaling and spine morphology. Encyclopedia of Neuroscience, p. 1141-1145.
Koskinen, M., Bertling, E., & Hotulainen, P. (2012). Methods to measure actin treadmilling rate in dendritic spines. In Methods in enzymology (Vol. 505, pp. 47-58). Academic Press.
Krasne F.B., Glanzman D.L. (1995). What we can learn from invertebrate learning. Annu Rev Psychol. 46:585.
Leuner, B., & Shors, T. J. (2010). Synapse formation and memory. In Encyclopedia of Behavioral Neuroscience (pp. 349-355). Elsevier Inc.
Mizumori S.J., Rosenzweig M.R., Bennett E.L. (1985). Long-term working memory in the rat: effects of hippocampally applied anisomycin. Behav Neurosci.;99:220. [PubMed]
Nicoll, R. A., & Kauer, J. A. Malenka. RC 1988. The current excitement in long-term potentiation. Neuron, 1, 97-103.
Park, P., Georgiou, J., Sanderson, T.M. et al. (2021). PKA drives an increase in AMPA receptor unitary conductance during LTP in the hippocampus. Nat Commun 12, 413. This article is licensed under a Creative Commons Attribution 4.0.
Rose S.P.R. (1992). Neuropsychology of Memory. Squire L.R., et al., editors. Guilford; New York: p. 547.
Rose S.P. (1995). Glycoproteins and memory formation. Behav Brain Res. 66:73. [PubMed]
Rosenzweig M.R., et al. (1992). Neuropsychology of Memory. Squire LR, et al., editors. Guilford; New York: p. 533.
Rosenzweig, M. R. (2007). Modification of brain circuits through experience. In Neural Plasticity and Memory: From Genes to Brain Imaging. CRC Press/Taylor & Francis, Boca Raton (FL); 2007. PMID: 21204433.
Serrano P.A., et al. (1994). Differential effects of protein kinase inhibitors and activators on memory formation in the 2-day-old chick. Behav Neural Biol. 61:60. [PubMed]
Traynelis S.F., Wollmuth L.P., McBain C.J., Menniti F.S., Vance K.M., Ogden K.K., Hansen K.B., Yuan H., Myers S.J., Dingledine, R. (2010). Glutamate receptor ion channels: structure, regulation, and function. Pharmacol. Rev. 62 (3): 405–496
Tully T, et al. A return to genetic dissection of memory in Drosophila. Cold Spring Harb Symp Quant Biol. 1996;61:207. [PubMed]
Yang, S., Yang, S., Moreira, T., Hoffman, G., Carlson, G. C., Bender, K. J., ... & Tang, C. M. (2014). Interlamellar CA1 network in the hippocampus. Proceedings of the National Academy of Sciences, 111(35), 12919-12924.
Additional References
Alvarez, V. A., & Sabatini, B. L. (2007). Anatomical and physiological plasticity of dendritic spines. Annu. Rev. Neurosci., 30, 79-97.
Bliss, T. V., & Lømo, T. (1973). Long‐lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path. The Journal of physiology, 232(2), 331-356.
Chidambaram, S. B., Rathipriya, A. G., Bolla, S. R., Bhat, A., Ray, B., Mahalakshmi, A. M., ... & Sakharkar, M. K. (2019). Dendritic spines: revisiting the physiological role. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 92, 161-193.
Eichenbaum, H. (1999). Conscious awareness, memory, and the hippocampus. Nature Neuroscience, 2 (9), 775–776.
Hayashi-Takagi, A., Yagishita, S., Nakamura, M., Shirai, F., Wu, Y.I., Loshbaugh, A.L., Kuhlman, B., Hahn, KM., Kasai, H. (2015). Labelling and optical erasure of synaptic memory traces in the motor cortex. Nature 525, 333–338.
Harris, K. M., Fiala, J. C., & Ostroff, L. (2003). Structural changes at dendritic spine synapses during long-term potentiation. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences, 358 (1432), 745-748.
Hebb, D.O. (1949). The Organization of Behavior. New York. Wiley.
Henry, F. E., Hockeimer, W., Chen, A., Mysore, S. P., & Sutton, M. A. (2017). Mechanistic target of rapamycin is necessary for changes in dendritic spine morphology associated with long-term potentiation. Molecular Brain, 10 (1), 1-17.
Jonides, J., Lacey, S. C., & Nee, D. E. (2005). Processes of working memory in mind and brain. Current Directions in Psychological Science, 14 (1), 2–5.
Kaada, B. R., Rasmussen, E. W., & Kveim, O. (1961). Effects of hippocampal lesions on maze learning and retention in rats. Experimental Neurology, 3(4), 333-355.
Kandel, E. (1976). Cellular Basis of Behavior. San Francisco. W.H. Freeman and Company.
Kandel, E. R., & Schwartz, J. H. (1982). Molecular biology of learning: Modulation of transmitter release. Science, 218 (4571), 433–443
Kasai, Haruo; Matsuzaki, Masanori; Noguchi, Jun; Yasumatsu, Nobuaki; Nakahara, Hiroyuki (1 July 2003). "Structure–stability–function relationships of dendritic spines". Trends in Neurosciences. 26 (7): 360–368.
Kasthuri, N., Hayworth, K. J., Berger, D. R., Schalek, R. L., Conchello, J. A., Knowles-Barley, S., ... & Lichtman, J. W. (2015). Saturated reconstruction of a volume of neocortex. Cell, 162 (3), 648-661.
Kim, C. H., & Lisman, J. E. (1999). A role of actin filament in synaptic transmission and long-term potentiation. Journal of Neuroscience, 19 (11), 4314-4324.
Kolb, B. & Whishaw, I.Q. (2001). In Introduction to Brain and Behavior. New York. Worth Publishers.
Krucker, T., Siggins, G. R., & Halpain, S. (2000). Dynamic actin filaments are required for stable long-term potentiation (LTP) in area CA1 of the hippocampus. Proceedings of the National Academy of Sciences, 97(12), 6856-6861.
Krupa, D. J., Thompson, J. K., & Thompson, R. F. (1993). Localization of a memory trace in the mammalian brain. Science, 260(5110), 989–991.
Lashley, K. S. (1929). The effects of cerebral lesions subsequent to the formation of the maze habit: Localization of the habit. In Brain mechanisms and intelligence: A quantitative study of injuries to the brain (pp. 86–108). Chicago, IL: University of Chicago Press.
Miller, R. R., & Marlin, N. A. (2014). Amnesia following electroconvulsive shock. Functional disorders of memory, 143-178. New York. Psychology Press.
Murakoshi, H., Wang, H., & Yasuda, R. (2011). Local, persistent activation of Rho GTPases during plasticity of single dendritic spines. Nature, 472 (7341), 100-104.
Ofer, N., Berger, D. R., Kasthuri, N., Lichtman, J. W., & Yuste, R. (2021). Ultrastructural analysis of dendritic spine necks reveals a continuum of spine morphologies. Developmental Neurobiology.
Pinel, P.J. & Barnes, S. (2021). Biopsychology (11th Edition). Boston. Pearson Education.
Sigurdsson, T., Doyère, V., Cain, C. K., & LeDoux, J. E. (2007). Long-term potentiation in the amygdala: A cellular mechanism of fear learning and memory. Neuropharmacology, 52 (1), 215–227.
Squire, L. R. (1992). Memory and the hippocampus: A synthesis from findings with rats, monkeys, and humans. Psychological Review, 99(2), 195–231.
Thompson, R. (1974). Localization of the “maze memory system” in the white rat. Physiological Psychology, 2 (1), 1-17.
Woodruff-Pak, D. S., Goldenberg, G., Downey-Lamb, M. M., Boyko, O. B., & Lemieux, S. K. (2000). Cerebellar volume in humans related to magnitude of classical conditioning. Neuroreport: For Rapid Communication of Neuroscience Research, 11(3), 609–615.
Yang, G., Pan, F., & Gan, W. B. (2009). Stably maintained dendritic spines are associated with lifelong memories. Nature, 462 (7275), 920-924.
Attributions
"Learning Objectives," "Overview," "The Search for Learning and Memory in the Synapse," "Changes at the Synapse Correlated with Learning and Memory," "Changes at the Synapse Correlated with Learning and Memory: Anatomical Changes in Dendritic Spines," "LTP, Neurochemical Cascades, and Stages of Memory," original material written by Kenneth A. Koenigshofer, Ph.D., is licensed under CC BY 4.0.
"Synaptic Plasticity," "Synaptic Plasticity: short-term enhancement, long-term potentiation and long-term depression," "Short-term Synaptic Enhancement and Depression," "Long-term Potentiation (LTP)," and "Long-term Depression (LTD)," adapted by Kenneth A. Koenigshofer, Ph.D., from Lumen Boundless Biology, How Neurons Communicate; https://courses.lumenlearning.com/bo...s-communicate/; curation and Revision. Provided by: Boundless.com. License: CC BY-SA: Attribution-ShareAlike.
"Cellular and Receptor Level Mechanisms Revisited" adapted from The Brain from Top to Bottom; license: Copyleft, https://thebrain.mcgill.ca/flash/pop.../pop_copy.html; modified by Kenneth A. Koenigshofer, PhD., licensed under CC BY 4.0.


