11.4: The Brain and Sleep
- Page ID
- 110609
This page is a draft and under active development. Please forward any questions, comments, and/or feedback to the ASCCC OERI (oeri@asccc.org).
- Describe the role of the pons, medulla, hypothalamus (SCN), thalamus, basal forebrain and pineal gland in the initiation and maintenance of sleep.
- Analyze the role of different neurochemicals, particularly the inhibitory neurotransmitter GABA and the sleep hormone melatonin in sleep initiation and maintenance.
- Contrast the role of other neurochemicals like excitatory neurotransmitters glutamate and norepinephrine in wakefulness and sleep.
Overview
In general, there are several regions of the brain that work in concert to increase wakefulness and arousal, or to cause us to fall asleep or stay asleep. The primary excitatory neurotransmitters like glutamate and acetylcholine and the primary inhibitory neurotransmitter GABA play a role in these processes.
During the Spanish flu of 1918 some patients went into coma and some were sleepless before they died. Von Economo did autopsies and found different types of lesions. He concluded that the posterior hypothalamus/upper part of midbrain might be a wakefulness center, and the preoptic area of anterior hypothalamus might be a sleep center. These areas are illustrated in Figure \(\PageIndex{1}\). Later countless autopsies suggest that when one goes into a coma, their brainstem has suffered damage.
Sleep is not a state of neural silence
Work by Moruzzi and Magoun (1949) also suggested that when the reticular formation of cats was destroyed, comas were triggered, and stimulating this part led to awakening from normal sleep. Also, since this part receives incoming messages via sensory pathways, they developed the concept of “ascending activating reticular system” or the prime contender for “wakefulness center.”
But these initial experiments had several issues. Since neurotoxic substances that were used to destroy the neurons/cell bodies of the posterior hypothalamus and reticular formation kept the axons that originated elsewhere intact, the wakefulness function that was diminished initially, quickly returned to normal.
Most importantly, these studies also showed that sleep is not a passive process, in which being deprived of sensory input causes sleep. Many people believe that sleep is something that happens in the absence of anything else like when someone is bored. For example, medical students attending a lecture in a subject that does not totally interest them will fall off to sleep. However, it is important to keep in mind that that is not actually what is happening. If a group of seven year old children who had had enough sleep the previous nights were sitting in the same lecture, they would not fall asleep but rather be bouncing off the walls!
In subsequent studies, application of electrical stimuli to the thalamus of cats when they were awake caused them to fall asleep. This indicates that sleep involves interactions between the thalamus and cortex. Also, when it was discovered that sleep and wakefulness cycles were not disrupted by sensory activation and that during REM there is intense activity in the cortex, the idea of sleep as passivity was further discredited.
There are two major neural circuits in the brainstem that operate in opposition to and alternation with each other. One of these circuits stimulates wakefulness, the other stimulates sleep, and their interaction is regulated by the body’s internal clock (SCN).
Some large areas of the cortex are thus under the control of these networks of small groups of neurons that are located in the brainstem and that form complex circuits, not single "sleep centers" or "wakefulness centers." Wakefulness, which is indispensable for survival, is thus ensured by a whole network of redundant structures.
Forebrain regions involved in sleep
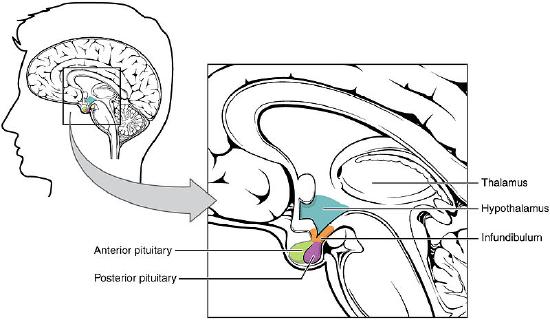
In the Figure \(\PageIndex{1}\) you can see the hypothalamus where the specific area of the brain, the suprachiasmatic nucleus (SCN- which is involved in the body's daily/circadian rhythm regulation) is located.
Anterior hypothalamus/SCN
SCN
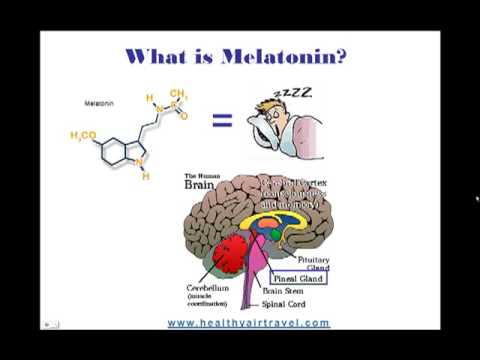
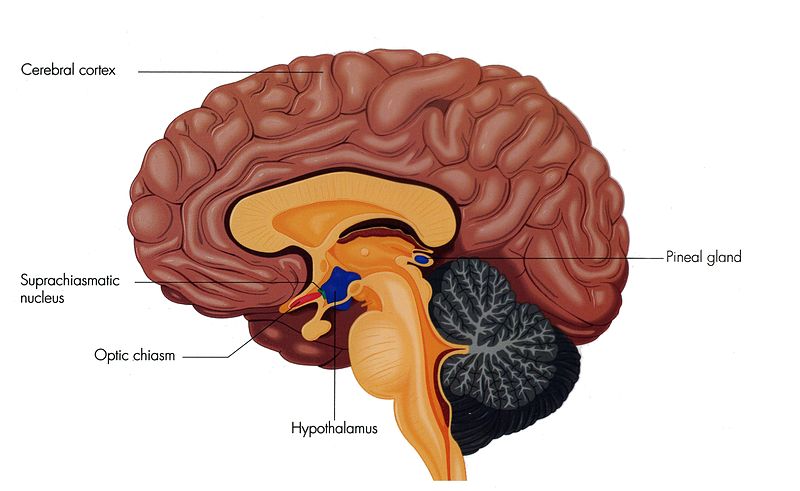
The SCN is considered "body's internal clock." It will be discussed in a later section in more detail. The SCN is a bilateral structure located in the anterior part of the hypothalamus. It is the central pacemaker of the circadian timing system and regulates most circadian rhythms in the body (Hastings et al, 2018). The SCN receives signals from many different places. The major one is the retinohypothalamic tract originating from photosensitive (light-sensitive) ganglion cells of the retina. The SCN sends signals to structures such as the pineal gland, producing melatonin during the night for induction of sleep. Disruptions in the SCN circadian system have been found to correlate with various mood disorders and sleep disorders.
Preoptic area
The anterior hypothalamus also plays a fundamental role in the process of falling asleep. This structure, and in particular its preoptic area, appears to be sensitive to the serotonin released during waking periods. Apparently, when serotonin stimulates this preoptic area of the anterior hypothalamus, its GABAergic (GABA producing) neurons in turn inhibit the posterior hypothalamus, thus encouraging sleep. Damage to these GABAergic neurons is known to cause insomnia, whereas stimulating them causes experimental subjects to fall asleep rapidly.
Sleep-wakefulness switch
In vitro experiments have indicated that the wake-promoting neurotransmitters serotonin, norepinephrine, and acetylcholine inhibit identified preoptic GABA neurons (Gallopin et al., 2000); therefore, mutually inhibitory interactions between the sleep-promoting preoptic region and the arousal-related hypothalamic and midbrain structures may provide a substrate for a “sleep–wakefulness switch” (McGinty and Szymusiak, 2000; Saper et al., 2001). Thus, activation of preoptic sleep-promoting cells could lead to sleep onset by inhibiting arousal structures; in turn, activation of arousal hypothalamic and midbrain structures could suppress activity by preoptic NREM sleep-promoting cells as well as REM-promoting neurons (Reinoso-Suárez et al., 2010) and facilitate the switch to wakefulness.
Posterior hypothalamus
Stimulating the posterior hypothalamus produces a state of wakefulness comparable to that induced by stimulating the reticular formation in the brainstem. The activity of the posterior hypothalamus diminishes naturally during sleep, when it releases less histamine, a molecule that it uses as a neurotransmitter. The antihistamines that people take for allergy symptoms are known to cause some sleepiness, by reducing the activity of histamine.
Thalamus
The thalamus contains neurons that send projections throughout the cortex. The activation of these thalamocortical neurons causes them to release excitatory amino acids such as aspartate and glutamate, thus contributing to excitation of the cortex and to wakefulness. During wakefulness, these neurons generate single action potentials at regular intervals. However, as the individual falls asleep these neurons begin firing in bursts instead, thus causing the cortex to display the synchronized EEG pattern that is typical of sleep (see section * for sleep stages).
Basal forebrain
The system of the basal forebrain is composed of neurons that synthesize acetylcholine and/or GABA. On their own, these neurons account for 70% of the cholinergic innervation of the cortex (where the cortex is activated by acetylcholine), while also sending projections to the thalamic nuclei. Stimulating these neurons causes wakefulness, but destroying them with neurotoxic substances causes wakefulness to decline for only a very short time.
In the situations just described, the cortical activation that causes wakefulness results from the direct stimulation of the cortical neurons by the various components of the wakefulness network. But these cortical neurons can also be activated in another way: by the inhibition of those neurons that naturally inhibit cortical activity. And that is exactly what the GABAergic neurons located in the posterior hypothalamus and the basal forebrain do: they inhibit other, cortical GABAergic neurons.
This executive network for wakefulness is itself activated by other systems arising in the brainstem.
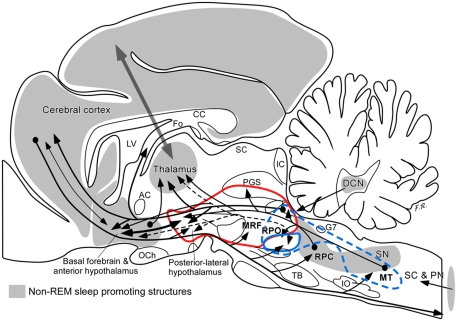
It is thus all of these wakefulness signals that stop reaching the cortex at the onset of non-REM sleep. They are interrupted at the thalamus, which serves as a true gatekeeper to the cortex and is greatly influenced by the diffuse neuromodulatory systems of the brainstem. The complexity of these interactions in NREM sleep is illustrated in the diagram of the cat brain below (See Figure \(\PageIndex{2}\)). This diagram illustrates a complex set of connections between cortical, subcortical, and brainstem structures that mediate various aspects of REM and NREM sleep. The figure is complicated, and the details are not important for our purpose here. It is included to illustrate the complexity of the connections between different brain sub-structures and signals being sent in order to maintain the individual's movement through the different stages of sleep throughout the night.
[A legend indicates that the thalamus–cerebral cortex complex or unit is darker to emphasize that these structures are necessary for the behavioral and bioelectric signs that characterize NREM sleep. AC, anterior commissure; CC, corpus callosum; DCN, deep cerebellar nuclei; Fo, fornix; G7, genu of the facial nerve; IC, inferior colliculus; IO, inferior olive; LV, lateral ventricle; MRF; midbrain reticular formation; MT, medullary tegmentum; OCh, optic chiasm; PGS, periaqueductal gray substance; RPC, caudal pontine reticular nucleus; RPO, oral pontine reticular nucleus; SC, superior colliculus; SC and PN, spinal cord and peripheral nerves; SN, solitary nucleus; TB, trapezoid body.]
The rhythmic activity pattern established by the thalamocortical neurons that disconnects the cortex from internal and external signals at the onset of non-REM sleep. In contrast, during the REM phases of sleep, the thalamus probably continues to pass at least some of these signals on to the cortex, at least in some fragmentary, filtered, or distorted form.
The regulation of wakefulness is essential for survival and involves several different redundant structures in the brain. None of these structures, taken in isolation, is indispensable for activation of the cortex. But three of these brain structures that send projections to the cortex are sufficient to maintain the desynchronized EEG pattern that is characteristic of wakefulness. These structures are the 1. posterior hypothalamus, 2. the (intralaminar nuclei of the) thalamus and 3. the basal forebrain. Together they are often referred to as the “executive network”.
Pineal Gland
Inferior and somewhat posterior to the thalamus is the pineal gland, a tiny endocrine gland whose functions are not entirely clear. The pinealocyte cells that make up the pineal gland are known to produce and secrete the amine hormone melatonin, which is derived from serotonin.
The secretion of melatonin varies according to the level of light received from the environment. When photons of light stimulate the retinas of the eyes, a nerve impulse is sent to the SCN. From the SCN, the nerve signal is carried to the spinal cord and eventually to the pineal gland, where the production of melatonin is inhibited. As a result, blood levels of melatonin fall, promoting wakefulness. In contrast, as light levels decline—such as during the evening—melatonin production increases, boosting blood levels and causing drowsiness. The pineal gland and melatonin are discussed in more detail in section *
Watch this video to view an animation describing the function of the hormone melatonin. What should you avoid doing in the middle of your sleep cycle that would lower melatonin?
[reveal-answer q=”598458″]Show Answer[/reveal-answer]
[hidden-answer a=”598458″]Turning on the lights.[/hidden-answer]
The secretion of melatonin may influence the body’s circadian rhythms (discussed in section *), the dark-light fluctuations that affect not only sleepiness and wakefulness, but also appetite and body temperature. Interestingly, children have higher melatonin levels than adults, which may prevent the release of gonadotropins from the anterior pituitary, thereby inhibiting the onset of puberty. Finally, an antioxidant role of melatonin is the subject of current research.
Jet lag occurs when a person travels across several time zones and feels sleepy during the day or wakeful at night. Traveling across multiple time zones significantly disturbs the light-dark cycle regulated by melatonin. It can take up to several days for melatonin synthesis to adjust to the light-dark patterns in the new environment, resulting in jet lag. Some air travelers take melatonin supplements to induce sleep.
Hindbrain regions involved in sleep
There are several "lower regions" of the brainstem that seem to be involved in sleep regulation according to early and current research.
Pons
The brainstem region known as the pons is critical for initiating REM sleep. During REM sleep, the pons sends signals to the visual nuclei of the thalamus and to the cerebral cortex (this region is responsible for most of our thought processes). The pons also sends signals to the spinal cord, causing the temporary paralysis that is characteristic of REM sleep. This is what happens in the experience of temporary sleep paralysis that many people might have experienced. Since the pons is effectively still "asleep" while the cortex is "awake," people experience the terrifying inability to move their body while they are completely conscious!
Also, the structures like the anterior hypothalamus play an important role in the onset of sleep.
Medulla and RAS
The reticular activating system (RAS) plays an important role in conscious awareness. According to Iwanczuk and Guzniczak (2015), "The ascending reticular activating system (ARAS) is responsible for a sustained wakefulness state. It receives information from sensory receptors of various modalities, .... [and those pathways] reach the thalamus directly or indirectly... The reticular activating system begins in the dorsal part of the posterior midbrain and anterior pons, continues into the diencephalon, and then divides into two parts reaching the thalamus and hypothalamus, which then project into the cerebral cortex."
The solitary tract nucleus region in the dorsal medulla is thought to provide a link between visceral activities such as respiratory, cardiovascular and gastrointestinal functions, and the sleep–wakefulness states. The solitary tract nucleus does not directly project to the cerebral cortex (Saper, 1995), although it does project to several brainstem, thalamic, and hypothalamic areas that innervate the cortex and can mediate EEG and sleep responses, such as the lateral hypothalamus, and nuclei of the midline thalamus.
Summary
The main brain regions working together to produce sleep and wakefulness have been discussed here. The neurons of the RAS play a role in arousal and sleep to some degree. The "executive network" seems to play an important role in regulating the responsiveness of the cortex to outside stimulation. The anterior hypothalamus and pineal regulate and fine tune many of these functions as well.
References
- Gallopin, T., Fort, P., Eggermann, E., Cauli, B., Luppi, P. H., Rossier, J., Audinat, E., Mühlethaler, M., & Serafin, M. (2000). Identification of sleep-promoting neurons in vitro. Nature, 404, 992–995.
- Hastings, M.H., Maywood, E.S., & Brancaccio, M. (2018). Generation of circadian rhythms in the suprachiasmatic nucleus. Nat Rev Neurosci. 19(8), 453-469.
- Iwańczuk, W., & Guźniczak, P. (2015). Neurophysiological foundations of sleep, arousal, awareness and consciousness phenomena. Part 1. Anaesthesiol Intensive Ther. 47(2), 162–167. doi:10.5603/AIT.2015.0015
- McGinty, D., and Szymusiak, R. (2000). The sleep-wake switch: a neuronal alarm clock. Nat. Med. 6, 510–511.
- Reinoso-Suárez, F., de la Roza, C., Rodrigo-Angulo, M. L., de Andrés, I., Núñez, A., and Garzón, M. (2010). “GABAergic mechanisms in the ventral oral pontine tegmentum – the REM sleep induction site- in the modulation of sleep-wake states,” in GABA and Sleep, ed. J. M. Monti, S. R. Pandi-Perumal, and H. Möhler (Basel: Birkhauser-Verlag), 233–252.
- Saper, C. B. (1995). “Central autonomic system,” in The Rat Nervous System, ed. G. Paxinos (San Diego: Academic Press), 107–135.
- Saper, C. B., Chou, T. C., and Scammell, T. E. (2001). The sleep switch: hypothalamic control of sleep and wakefulness. Trends Neurosci. 24, 726–731.
Attribution
- The Brain structures that wake you up and put you to sleep by Bruno Dubuc under a Copyleft license.
- Functional Anatomy of Non-REM Sleep by Isabel de Andrés, Miguel Garzón and Fernando Reinoso-Suárez in Frontiers Neuroscience licensed CC-BY 4.0
- Information about sleep by NIH is in the public domain
- The Pineal Gland by Libretexts has no license indicated.
- Neuroanatomy, Nucleus Suprachiasmatic by Melinda A. Ma and Elizabeth H. Morrison licensed CC-BY 4.0