4.3: Nervous System Development
- Page ID
- 142136
This page is a draft and under active development. Please forward any questions, comments, and/or feedback to the ASCCC OERI (oeri@asccc.org).
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Explain how the neural tube forms
- Describe the growth and differentiation of the anterior neural tube into primary and secondary vesicles
- Understand the mechanisms of postnatal brain development
- Describe the development of the posterior neural tube into the adult spinal cord
- Identify at least four stages of neuron development
Overview
This module starts with the value of an embryologic perspective, and then discusses the formation of the neural tube, embryonic brain development (the process of the anterior neural tube differentiates into primary and secondary vesicles), postnatal brain development, spinal cord development, and neuron development.
An Embryologic Perspective
The brain is a complex organ composed of gray and white matter, which can be hard to distinguish. Starting from an embryologic perspective allows you to understand more easily how the parts relate to each other. The embryonic nervous system begins as a very simple structure—essentially a plate of tissue, which then gets increasingly complex. Looking at the development of the nervous system through a few early snapshots makes it easier to understand the whole complex system. Many structures that appear to be adjacent in the adult brain are not connected, and the connections that exist may seem arbitrary. But there is an underlying order to the system that comes from how different parts develop. By following the developmental pattern, it is possible to distinguish the major regions of the nervous system.
Neural Tube
To begin, a sperm cell and an egg cell fuse to become a fertilized egg. The fertilized egg cell, or zygote, starts dividing to generate the cells that make up an entire organism. Sixteen days after fertilization, the developing embryo’s cells belong to one of three germ layers that give rise to the different tissues in the body (Figure \(\PageIndex{1}\)). The endoderm, or inner tissue, is responsible for generating the lining tissues of various spaces within the body, such as the mucosae of the digestive and respiratory systems. The mesoderm, or middle tissue, gives rise to most of the muscle and connective tissues. Finally the ectoderm, or outer tissue, develops into the integumentary system (the skin) and the nervous system. It is probably not difficult to see that the outer tissue of the embryo becomes the outer covering of the body. But how is it responsible for the nervous system?
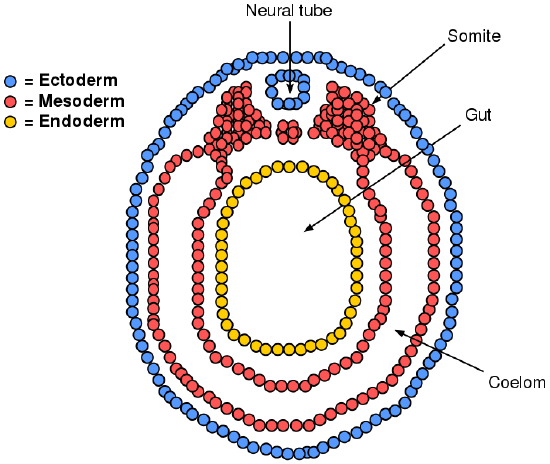
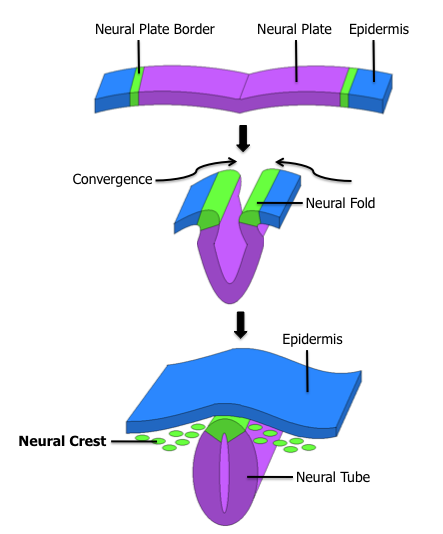
As the embryo develops, a portion of the ectoderm differentiates into tissue that will become the nervous system. Molecular signals induce cells in this region to form a neural plate. The cells then begin to change shape, causing the tissue to buckle and fold inward (Figure \(\PageIndex{2}\)). A neural groove forms, visible as a line along the dorsal surface of the embryo. The ridge-like edge on either side of the neural groove is referred to as the neural fold. As the neural folds come together and converge, the underlying structure forms into a tube called the neural tube, located just beneath the remaining ectoderm (which will form the epidermis).
At this point, the early nervous system is a simple, hollow tube. It runs from the anterior end of the embryo to the posterior end. Beginning at 25 days, the anterior end develops into the brain, and the posterior portion becomes the spinal cord. This is the most basic arrangement of tissue in the nervous system, and by the fourth week of development it gives rise to the more complex structures.
Brain Development
Embryonic Brain Development
Overview
The vertebrate brain has three major regions based on embryonic development: the forebrain (including the cerebrum, thalamus, hypothalamus, and limbic system structures), the midbrain, and the hindbrain (including the medulla, pons, and cerebellum). Figure \(\PageIndex{3}\) shows the embryologic vesicles (originating from bulges in the neural tube, explained in greater detail below) that contribute to these regions. The prosencephalon (forebrain) is composed of the telencephalon and the diencephalon, the mesencephalon is the midbrain, and the rhombencephalon (hindbrain) is composed of the metencephalon and the myelencephalon. The spinal cord extends below the hindbrain.
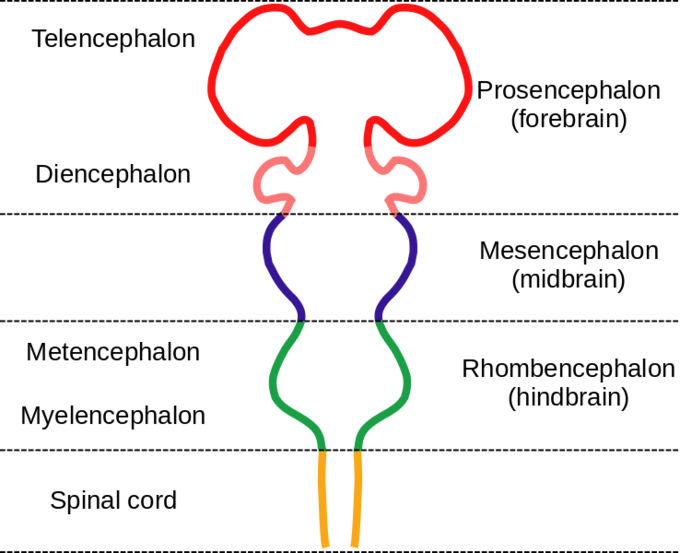
The diencephalon is the one region of the adult brain that retains its (Greek) embryonic name in common usage. This is because there is no better term for it (dia = “through”). The diencephalon is between the cerebrum and the rest of the nervous system and can be described as the region through which all projections have to pass. All vertebrate brains have these regions, but in humans the anterior forebrain is enlarged to the extent that the diencephalon (posterior forebrain), the midbrain, and even part of the hindbrain is hidden from view by the cerebrum.
Another aspect of the adult CNS structures that relates to embryonic development is the cerebral ventricles—open spaces within the CNS where cerebrospinal fluid circulates. They are the remnant of the hollow center of the neural tube. The four ventricles and the tubular spaces associated with them can be linked back to the hollow center of the embryonic brain.
Primary Vesicles
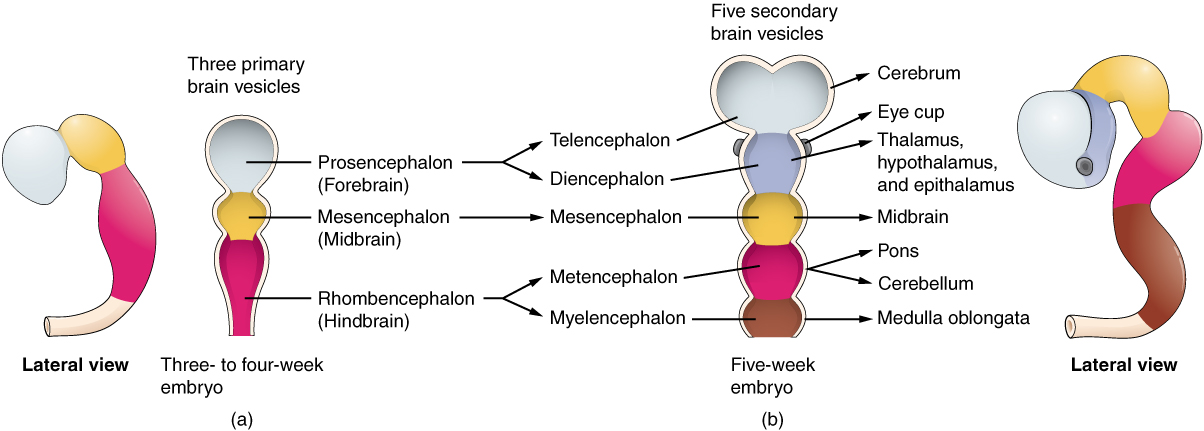
When the embryo is three to four weeks of age, the anterior end of the neural tube starts to develop into the brain. It undergoes a couple of enlargements; the result is the production of sac-like vesicles. Similar to a child’s balloon animal, the long, straight neural tube begins to take on a new shape. Three vesicles form at the first stage, which are called primary vesicles. These vesicles are given names that are based on Greek words, the main root word being enkephalon, which means “brain” (en- = “inside”; kephalon = “head”). The prefix to each generally corresponds to its position along the length of the developing nervous system.
The prosencephalon (pros- = “in front”) is the forward-most vesicle, and the term can be loosely translated to mean forebrain. The mesencephalon (mes- = “middle”) is the next vesicle, which can be called the midbrain. The third vesicle at this stage is the rhombencephalon. The first part of this word is also the root of the word rhombus, which is a geometrical figure with four sides of equal length (a square is a rhombus with 90° angles). Whereas prosencephalon and mesencephalon translate into the English words forebrain and midbrain, there is not a word for “four-sided-figure-brain.” Instead, the third vesicle is called the hindbrain. One way of thinking about how the brain is arranged is to use these three regions—forebrain, midbrain, and hindbrain—which are based on the primary vesicle stage of development (Figure \(\PageIndex{4}\)a).
Secondary Vesicles
The brain continues to develop in a five-week old embryo, and the vesicles differentiate further (Figure \(\PageIndex{4}\)b). The three primary vesicles become five secondary vesicles. The prosencephalon enlarges into two new vesicles called the telencephalon and the diencephalon. The telecephalon will become the cerebrum. The diencephalon gives rise to several adult structures; two that will be important are the thalamus and the hypothalamus.
The mesencephalon does not differentiate any further, but remains an established region of the brain. The rest of the brain develops around it and constitutes a large percentage of the mass of the brain. Dividing the brain into forebrain, midbrain, and hindbrain is useful in considering its developmental pattern, but the midbrain is a relatively small proportion of the entire brain.
The rhombencephalon develops into the metencephalon and myelencephalon. The metencephalon corresponds to the adult structure known as the pons and also gives rise to the cerebellum. The cerebellum (from the Latin meaning “little brain”) accounts for about 10 percent of the mass of the brain and is an important structure in itself. The most significant connection between the cerebellum and the rest of the brain is at the pons, because the pons and cerebellum develop out of the same vesicle. The myelencephalon corresponds to the adult structure known as the medulla (or medulla oblongata).
Postnatal Brain Development
As summarized by Stiles & Jernigan (2010, p. 328), "Human brain development is a protracted process that begins in the third gestational week ... and extends at least through late adolescence, arguably throughout the lifespan". During the preschool years (roughly age three to five years), the brain quadruples in size, attaining about 90% of adult volume by age six (Stiles & Jernigan, 2010). Figure \(\PageIndex{5}\) illustrates the growth between a one-month old baby and a six-year-old child, in both brain size and neuron complexity.
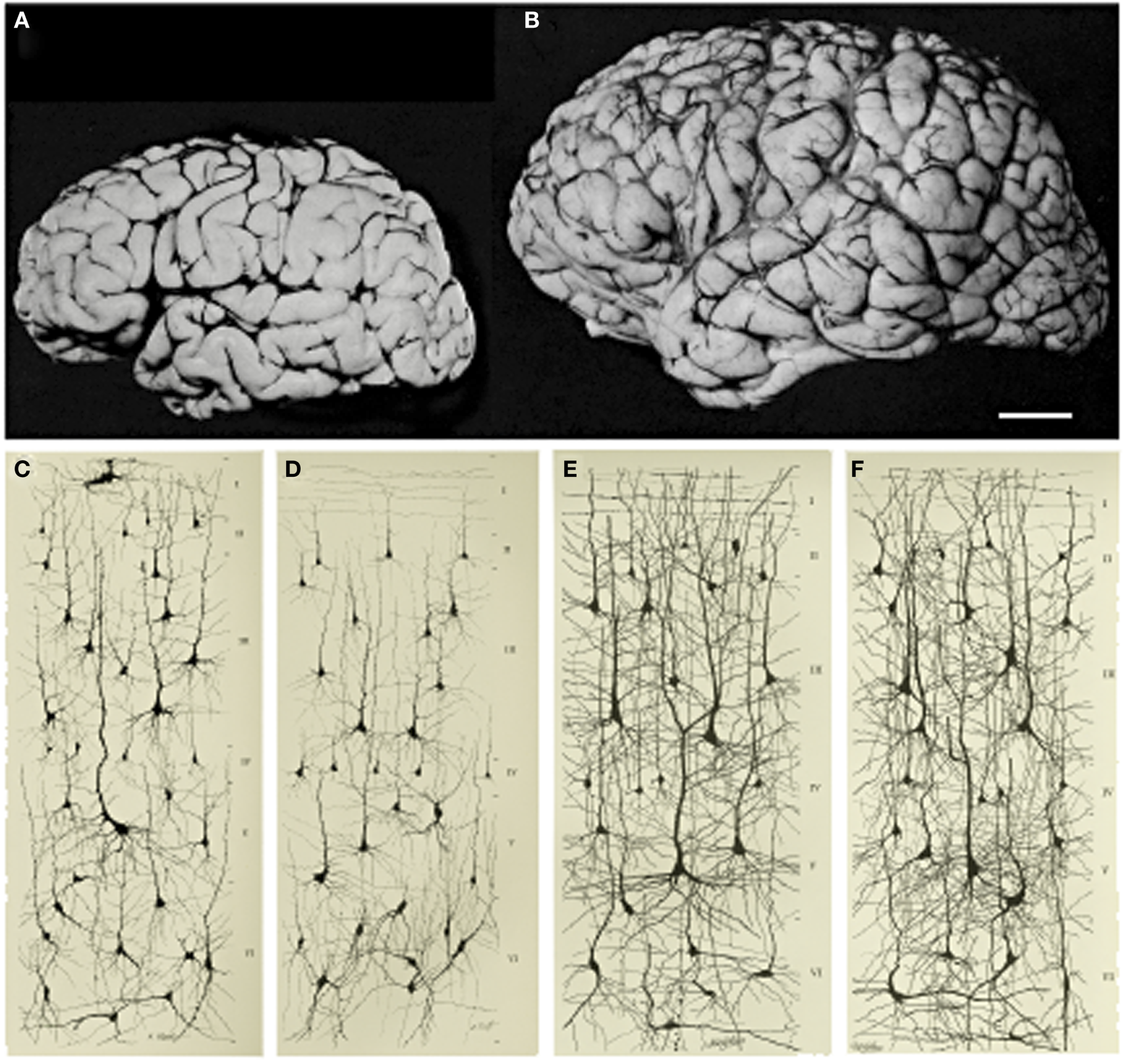
Changes in the structure of both gray matter and white matter components of the brain continue through childhood and adolescence, accompanied by changes in both functional organization and behavior. Young children, particularly infants, have much higher connectivity between neurons in the brain than adults. As development continues, these connections become more specialized (through pruning of less efficient pathways, as determined by experience- see neuron development below). Correspondingly, "plasticity and capacity for adaptation ... is the hallmark of early brain development" (Stiles & Jernigan, 2010, p. 328). Plasticity (the ability of the brain to change based on experience) continues throughout life, but slows down with age. It is still possible to "teach an old dog new tricks"- it just takes longer!
Although the human brain increases five-fold in volume between infancy and adulthood, there is little change in the number of neurons present. Brain growth occurs as a result of axon myelination and increased connections between neurons (Budday et al., 2015).
Spinal Cord Development
While the brain is developing from the anterior neural tube, the spinal cord is developing from the posterior neural tube. However, its structure does not differ from the basic layout of the neural tube. It is a long, straight cord with a small, hollow space down the center. The neural tube is defined in terms of its anterior versus posterior portions, but it also has a dorsal–ventral dimension. As the neural tube separates from the rest of the ectoderm, the side closest to the surface is dorsal (toward the back), and the deeper side is ventral (toward the belly).
As the spinal cord develops, the cells making up the wall of the neural tube proliferate and differentiate into the neurons and glia of the spinal cord. The dorsal tissues will be associated with sensory functions, and the ventral tissues will be associated with motor functions
Development of the Nervous System: Spina Bifida
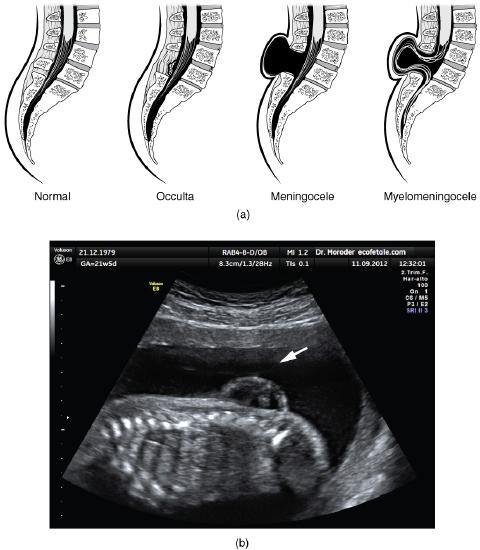
Early formation of the nervous system depends on the formation of the neural tube. A groove forms along the dorsal surface of the embryo, which becomes deeper until its edges meet and close off to form the tube. If this fails to happen in the posterior region where the spinal cord forms, a developmental defect called spina bifida occurs. The closing of the neural tube is important for more than just the proper formation of the nervous system. The surrounding tissues are dependent on the correct development of the tube. The connective tissues surrounding the CNS can be involved as well.
There are three classes of this disorder: occulta, meningocele, and myelomeningocele (Figure \(\PageIndex{6}\)). The first type, spina bifida occulta, is the mildest because the vertebral bones do not fully surround the spinal cord, but the spinal cord itself is not affected. No functional differences may be noticed, which is what the word occulta means; it is hidden spina bifida. The other two types both involve the formation of a cyst—a fluid-filled sac of the connective tissues that cover the spinal cord called the meninges. “Meningocele” means that the meninges protrude through the spinal column but nerves may not be involved and few symptoms are present, though complications may arise later in life. “Myelomeningocele” means that the meninges protrude and spinal nerves are involved, and therefore severe neurological symptoms can be present.
Often surgery to close the opening or to remove the cyst is necessary. The earlier that surgery can be performed, the better the chances of controlling or limiting further damage or infection at the opening. For many children with meningocele, surgery will alleviate the pain, although they may experience some functional loss. Because the myelomeningocele form of spina bifida involves more extensive damage to the nervous tissue, neurological damage may persist, but symptoms can often be handled. Complications of the spinal cord may present later in life, but overall life expectancy is not reduced.
Neuron Development
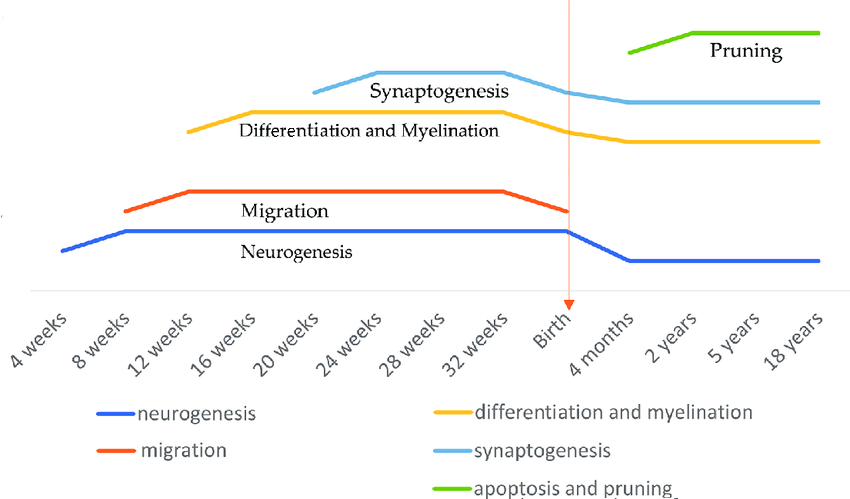
Neuron development is typically divided into several stages, although the names of the stages may vary somewhat from one source to another. Neuron production (also called proliferation) is the first stage, when neurogenesis, the production of new neurons from stem cells, occurs (Kalat, 2019). In humans, this begins around the fifth week of gestation (Budday et al., 2015), or on the 42nd day after conception (Stiles & Jernigan, 2010). It is mostly finished by 28 weeks gestational age, and if premature birth occurs before 28 weeks, further neurogenesis is inhibited (Kalat, 2019). Figure \(\PageIndex{7}\) shows a photograph of neurons developing in an embryo.
As the neurons are forming, migration (movement to their final destinations) starts, following chemical signals (Kalat, 2019). The six-layered mature structure of the cerebral cortex is formed via the orderly migration of neurons (Stiles & Jernigan, 2010). Differentiation is the process of a neuron achieving the features that make it distinct from other body cells- forming an axon and one or more dendrites (Kalat, 2019). The term differentiation also encompasses the formation of different types of neurons (Stiles & Jernigan, 2010), or gaining the features that distinguish one type of neuron from another.
Synaptogenesis is the creation of connections between neurons by forming synapses (Kalat, 2019). It begins mid-gestation, and as connectivity progresses, axons form and reach out to numerous targets "until each neuron connects with thousands of other neurons" by birth (Budday et al., 2015, p. 5). Synaptogenesis "continues throughout life, as neurons form new synapses and discard old ones" (Kalat, 2019, p. 118).
Myelination is the process of axons becoming insulated with a layer of myelin, which speeds up the transmission of nerve impulses. Myelination starts in the spinal cord and then progresses from the hindbrain to the midbrain and finally the forebrain. This occurs gradually over decades and may also be implicated in learning new motor skills (Kalay, 2019).
Synaptic pruning is the process of removing synapses that are not useful or efficient, based on the specific experiences of the individual. Pruning starts around birth and is completed during adolescence, by the time sexual maturity is attained. It is thought that learning corresponds with pruning (Budday et al., 2015).
An estimated timeline for these stages of neuron development is shown in Figure \(\PageIndex{8}\).
Summary
The development of the nervous system starts early in embryonic development. The outer layer of the embryo, the ectoderm, gives rise to the skin and the nervous system. A specialized region of this layer becomes a groove that folds in and becomes the neural tube beneath the dorsal surface of the embryo. The anterior end of the neural tube develops into the brain, and the posterior region becomes the spinal cord.
The brain develops from this early tube structure and gives rise to specific regions of the adult brain. As the neural tube grows and differentiates, it enlarges into three vesicles that correspond to the forebrain, midbrain, and hindbrain regions of the adult brain. Later in development, two of these three vesicles differentiate further, resulting in five vesicles. Those five vesicles can be aligned with the four major regions of the adult brain. The cerebrum is formed directly from the telencephalon. The diencephalon is the only region that keeps its embryonic name, and includes the thalamus and the hypothalamus. The mesencephalon becomes the midbrain, the metencephalon forms the pons and the cerebellum, and the myelencephalon becomes the medulla.
Brain development is a lifelong process, and the brain retains plasticity (the ability to change based on experience) throughout life. The increase in brain size during postnatal brain development occurs largely due to the myelination of axons and increased connections between neurons.
The spinal cord develops from the remainder of the neural tube and retains the tube structure, with the nervous tissue thickening and the hollow center becoming a very small central canal through the cord. The rest of the hollow center of the neural tube corresponds to open spaces within the brain called the ventricles, where cerebrospinal fluid is found.
Several stages of neuron development have been identified- neuron production (or proliferation), migration, differentiation, synaptogenesis (increased connectivity), myelination, and synaptic pruning.
References
Budday, S., Steinmann, P., & Kuhl, E. (2015). Physical biology of human brain development. Frontiers in Cellular Neuroscience 9, 1-17. DOI=10.3389/fncel.2015.00257
Kalat, J.W. (2019). Biological Psychology (13th ed.). Cengage.
Stiles, J. & Jernigan, T.L. (2010). The basics of brain development. Neuropsychology Review 20, 327–348. DOI 10.1007/s11065-010-9148-4
Attributions
- Figures:
- Vertebrate embryo by Jlesk is licensed under CC BY-SA 3.0, via Wikimedia Commons
- Neural crest via Wikimedia Commons has been released into the public domain by its author, Abitua at English Wikipedia. This applies worldwide.
- Embryologic Brain Vesicles- no specific attribution (from " Development of the Human Brain" by LibreTexts, licensed under notset). Note: Text from this source is imprecise, sometimes to the point of being inaccurate, and is not included in the text adaptation on this page.
- Brain Vesicle Development by OpenStax is licensed under CC BY 4.0, via Wikimedia Commons
- Brain maturation by Javier DeFelipe is licensed CC BY 3.0, via Wikimedia Commons
- Spina Bifida by OpenStax is licensed under CC BY-SA 3.0 / Ultrasound image: "Spina bifida lombare sagittale" by Wolfgang Moroder is licensed under CC BY-SA 3.0; both are via Wikimedia Commons
- Neuron cluster by M. Oktar Guloglu, CC BY-SA 4.0, via Wikimedia Commons
- Timeline of neuron development adapted by Naomi Bahm (spelling corrected) from Timeline of brain development by Merve Çikili Uytun is licensed under CC BY 3.0, via Çikili Uytun, Merve. (2018). Development Period of Prefrontal Cortex. (From Prefrontal Cortex, edited by Ana Starcevic and Branislav Filipovic.) http://dx.doi.org/10.5772/intechopen.78697
- Text adapted from:
- Changes: Text (and some of the images) from above source pieced together with some modifications, transitions and additional content and images (particularly the embryonic brain development overview, and sections on postnatal brain development, and neuron development) added by Naomi I. Gribneau Bahm, PhD., Psychology Professor at Cosumnes River College, Sacramento, CA.


