9.1.5: The Genus Australopithecus
- Last updated
- Save as PDF
- Page ID
- 136430
The Australopithecines are a diverse group of hominins comprised of various species. Australopithecus is the given group or genus name. It stems from the Latin word Australo, meaning “southern,” and the Greek word pithecus, meaning “ape.” Within this section, we will outline these differing species’ geological and temporal distributions across Africa, unique derived and/or shared traits, and importance in the fossil record.
Between 3 mya and 1 mya, there seems to be differences in dietary strategy between species of hominins designated as Australopithecines, which is evident from the peculiar size of the molars in one of the groups. This pattern of larger posterior dentition (even relative to the incisors and canines), thick enamel, and cranial evidence for large chewing muscles is far more pronounced in a group known as the robust australopithecines, as opposed to their earlier contemporaries or predecessors, the gracile australopithecines, and certainly larger than those seen in early Homo, which emerges during this time. This pattern of incredibly large hind dentition (and very small anterior dentition) has led people to refer to robust australopithecines as megadont hominins (Figure 9.10).
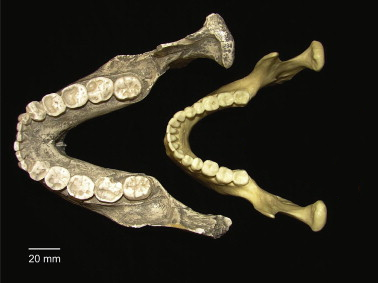 Figure \(\PageIndex{1}\): Robust Australopithecines (left) had large molars and chewing muscles compared to modern humans (right).
Figure \(\PageIndex{1}\): Robust Australopithecines (left) had large molars and chewing muscles compared to modern humans (right).This section has been categorized into “gracile” and “robust” Australopithecines, highlighting the morphological differences between the two groups (which many researchers have designated as separate genera: Australopithecus and Paranthropus, respectively) and then focusing on the individual species. It is worth noting, however, that not all researchers accept these clades as biologically or genetically disparate, with some researchers insisting that the relative gracile and robust features found in these species are due to parallel evolutionary events, toward similar dietary niches.
Despite this genus’ ancestral traits and small cranial capacity, all members show evidence of bipedal locomotion. It is generally accepted that Australopithecus species display varying degrees of arborealism and bipedality—these individuals were walking on the ground on two legs but were probably still comfortable with climbing trees.
Gracile Australopithecines
The section below describes individual species from across Africa. These species have coined the term “gracile australopithecines” because of the less exaggerated, smaller, and less robust features seen in the divergent “robust” group. Numerous Australopithecine species have been named, but some are still only based on a handful of fossil finds, whose designations are controversial.
East African Australopithecines
East African Australopithecines are found throughout the EARS, a system running from Malawi to Ethiopia, and include the earliest species associated with this genus. Numerous fossil-yielding sites, such as Olduvai, Turkana, and Laetoli, have excellent, datable stratigraphy, owing to the layers of volcanic tufts that have accumulated over millions of years. These tufts may be dated using absolute dating techniques, such as Potassium-Argon dating. This means that it is possible to know a relatively refined date for any fossil if the context of that find is known. Similarly, comparisons between the faunal assemblages of these stratigraphic layers have allowed researchers to chronologically identify environmental changes.
The earliest known Australopithecine is dated to 4.2 mya to 3.8 mya. Australopithecus anamensis (after “Anam,” meaning “lake” from the Turkana region in Kenya; Leakey et al. 1995; Patterson and Howells 1967) is currently found from sites in the Turkana region (Kenya) and Middle Awash (Ethiopia; Figure 9.11). Recently, a 2019 find from Ethiopia, named MRD, after Miro Dora where it was found, was discovered by an Ethiopian herder named Ali Bereino. It is one of the most complete cranial finds of this species (Ward et al. 1999). A small brain size (370 cc), relatively large canines, projecting cheekbones, and primitive earholes show more primitive features as compared to those of more recent Australopithecines. The most important element discovered associated with this species that indicates bipedalism is a fragment of a tibia (shinbone), which demonstrates features associated with weight transfer during bipedal walking. Similarly, the earliest found hominin femur belongs to this species. Primitive traits in the upper limb (such as the humerus) indicate some retained arboreal locomotion. Some researchers suggest that Au. anamensis is an intermediate form of the chronospecies that becomes Au. afarensis, evolving from Ar. ramidus. However, this is debated, with other researchers suggesting morphological similarities and affinities with more recent species instead. Almost 100 specimens, representing over 20 individuals, have been found to date (Leakey et al. 1995; McHenry, 2009; Ward et al. 1999).
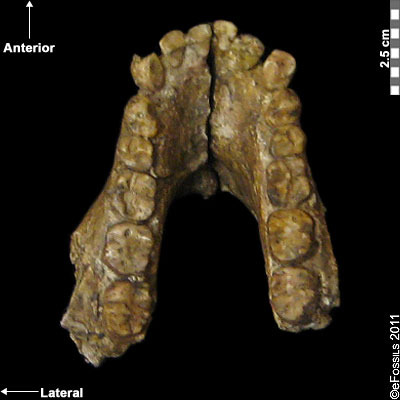 Figure \(\PageIndex{2}\): As seen in this mandible, Australopithecus anamensis had relatively large canine teeth.
Figure \(\PageIndex{2}\): As seen in this mandible, Australopithecus anamensis had relatively large canine teeth.Au. afarensis is one of the oldest and most well-known australopithecine species and consists of a large number of fossil remains. Au. afarensis (which means “from the Afar region”) is dated to between 2.9 mya and 3.9 mya and is found in sites all along the EARS system, in Tanzania, Kenya, and Ethiopia (Figure 9.12). The most famous individual stemming from this species is a partial female skeleton discovered in Hadar (Ethiopia), which was later nicknamed “Lucy,” after the Beatles’ song “Lucy in the Sky with Diamonds,” which was played in celebration of the find (Johanson et al. 1978; Kimbel and Delezene 2009). This skeleton was found in 1974 by Donald Johanson and dates to approximately 3.2 mya (Figure 9.13). In addition, in 2002 a juvenile of the species was found by Zeresenay Alemseged and given the name “Selam” (meaning “peace,” DIK 1-1), though it is popularly known as “Lucy’s Child” or as the “Dikika Child”(Alemseged et al. 2006). Similarly, the “Laetoli Footprints” (discussed in Chapter 7; Hay and Leakey 1982; Leakey and Hay 1979) have drawn much attention.
 Figure \(\PageIndex{3}\): Artistic reconstructions of Australopithecus afarensis by artist John Gurche. Female “Lucy” is left and a male is on the right.
Figure \(\PageIndex{3}\): Artistic reconstructions of Australopithecus afarensis by artist John Gurche. Female “Lucy” is left and a male is on the right.The canines and molars of Au. afarensis are reduced relative to great apes but are larger than those found in modern humans (indicative of a generalist diet); in addition, Au. afarensis has a prognathic face (the face below the eyes juts anteriorly) and robust facial features that indicate relatively strong chewing musculature (compared with Homo) but which are less extreme than in Paranthropus. Despite a reduction in canine size in this species, large overall size variation indicates high levels of sexual dimorphism in this species.
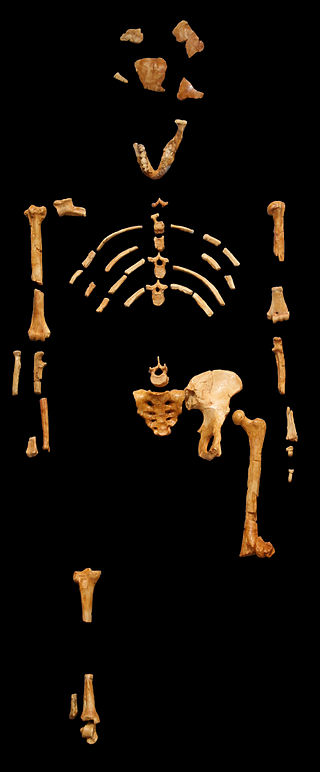 Figure \(\PageIndex{4}\): The humanlike femoral neck, valgus knee, and bowl-shaped hip seen in the “Lucy” skeleton indicates that Australopithecus afarensis was bipedal.
Figure \(\PageIndex{4}\): The humanlike femoral neck, valgus knee, and bowl-shaped hip seen in the “Lucy” skeleton indicates that Australopithecus afarensis was bipedal.Skeletal evidence indicates that this species was bipedal, primarily through examining the pelvis and lower limb, which demonstrate a humanlike femoral neck, a valgus knee, and bowl-shaped hip. More evidence of bipedalism is found not in the skeleton but in the footprints of this species. Au. afarensis is associated with the Laetoli Footprints, a 24-meter trackway of hominin fossil footprints preserved in volcanic ash discovered by Mary Leakey in Tanzania and dated to 3.5 mya to 3 mya. This set of prints is thought to have been produced by three bipedal individuals as there are no knuckle imprints, no opposable big toes, and a clear arch is present. The infants of this species are thought to have been more arboreal than the adults, as was discovered through analyses of the foot bones of the Dikika Child dated to 3.32 mya (Alemseged et al. 2006).
Although not found in direct association with stone tools, potential evidence for cut marks on bones, found at Dikika, and dated to 3.39 mya indicates a potential temporal/geographic overlap between meat eating, tool use, and this species. However, this evidence is fiercely debated. Others have associated the cut marks with the earliest tool finds from Lomekwi, Kenya, temporally (3.3 mya) and in close geographic proximity to this species.
Contested Species
Many named species are highly debated and/or newly described. Often these species are argued to have specimens associated with a more variable Au. afarensis or Au. anamensis species. Sometimes these specimens from which these species are named are dated to times, or found in places, when there are “gaps” in the palaeoanthropological record. Often these are argued to represent chronospecies or variants of Au. afarensis. However, it is possible that, with more discoveries, these species definitions will hold.
Australopithecus bahrelghazali (named after the Bahr el Ghazal river valley in the southern region of Borkou-Ennedi Tibesti in Chad, which is not within the EARS system) is dated to within the time period of Au. afarensis (3.6 mya; Brunet et al. 1995). Yet this discovery holds considerable significance in the field as this was the first Australopithecine to be discovered in Chad in central Africa (unlike those in the usual east Africa and South Africa). Researchers argue that the holotype, whom discoverers have named “Abel,” falls under the range of variation of Au. afarensis and therefore that A. bahrelghazali does not fall into a new species (Lebatard et al. 2008). If “Abel” is a member of Au. afarensis, the geographic range of the species would be extended even further.
On a different note, Australopithecus deyiremada (meaning “close relative” in the Ethiopian language of Afar) is dated to 3.5 mya to 3.3 mya and is based on fossil mandible bones discovered in 2011 in Woranso-Mille (in the Afar region of Ethiopia) by Yohannes Haile-Selassie, an Ethiopian paleoanthropologist (Haile-Selassie et al. 2019). The discovery indicated smaller teeth with thicker enamel than seen in Au. afarensis (potentially suggesting a harder diet) and a larger mandible and more projecting cheekbones than in Au. afarensis. The discoverers believe that this was a pivotal find in the palaeoanthropology field as it shows evidence of more than one closely related hominin species occupying the same region at the same temporal period (Haile-Selassie et al. 2015; Spoor 2015). If so, it may also imply that other Au. afarensis specimens have been incorrectly designated and that researchers should attempt to better understand the variability among these species. However, others have argued that this species has been prematurely identified and that more evidence is needed before splitting the taxa, since the variation appears subtle and may be due to slightly different niche occupations between populations over time.
Australopithecus garhi is another species found in the Middle Awash region of Ethiopia. It is currently dated to 2.5 mya and is therefore younger than Au. afarensis. Researchers have suggested it fills in a much-needed temporal “gap” between hominin finds in the region. It has a relatively large cranial capacity (450 cc) and larger hind dentition than seen in other gracile Australopithecines. The discoverers argue that the postcrania is also different (Afsaw et al. 1999), and the femur of a fragmentary partial skeleton (argued to belong to Au. garhi) indicates that this species may be longer limbed than Au. afarensis, although still able to move arboreally. However, this species is not well documented or understood and is based on only several fossil specimens. More astonishingly, crude/primitive stone tools resembling Oldowan (which will be described later) have been found in association with Au. garhi. While lacking some of the features of the Oldowan, this is one of the earliest technologies found in direct association with a hominin.
Kenyanthopus platyops (the name “platyops” refers to its flatter-faced appearance) is a highly contested genus/species designation of a specimen (KNM-WT 40000) from Lake Turkana in Kenya, discovered by Maeve Leakey in 1999 (Figure 9.14). Dated to between 3.5 mya and 3.2 mya, some have suggested this specimen is an Australopithecus, perhaps even Au. afarensis (with a brain size which is difficult to determine, yet appears small), while still others have placed this specimen in Homo (small dentition and flat-orthognathic face). While taxonomic placing of this species is quite divided, the discoverers have argued that this species is ancestral to Homo, in particular to Homo ruldolfensis (Leakey et al. 2001). Some have placed the species itself into the genus Homo, although the cranial capacity and general cranial features are not as derived. Some researchers have additionally associated the earliest tool finds from Lomekwi, Kenya, temporally (3.3 mya) and in close geographic proximity to this species/specimen.
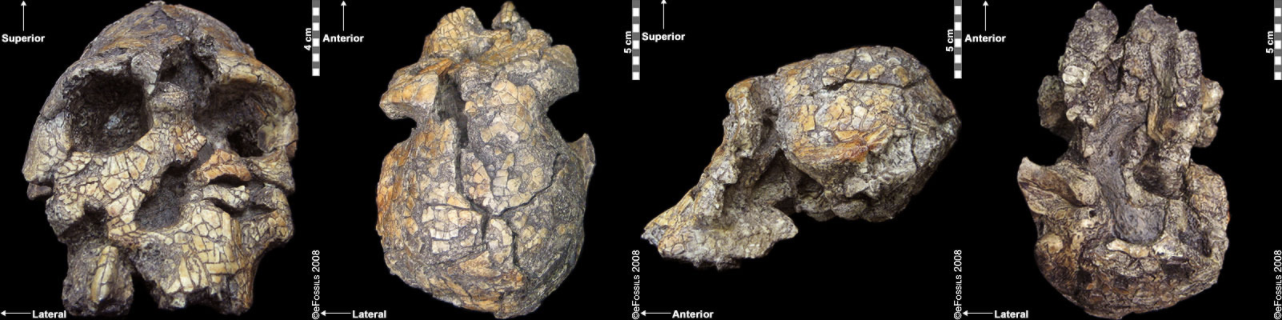 Figure \(\PageIndex{5}\): This specimen (Kenyanthopus platyops) has small detention, a small brain case, and a relatively flat face. Its genus/species designation remains contested.
Figure \(\PageIndex{5}\): This specimen (Kenyanthopus platyops) has small detention, a small brain case, and a relatively flat face. Its genus/species designation remains contested.South African Australopithecines
Since the discovery of the Taung Child, there have been numerous Australopithecine discoveries from the region known as “The Cradle of Humankind,” recently given UNESCO World Heritage Site status as “The Fossil Hominid Sites of South Africa.” The limestone caves found in the Cradle allow for the excellent preservation of fossils. Past animals navigating the landscape and falling into cave openings, or caves used as dens by carnivores, led to the accumulation of deposits over millions of years. Many of the hominin fossils we have, encased in breccia (hard, calcareous sedimentary rock), are recently exposed from limestone quarries that were mined in the previous century. This means that extracting fossils requires excellent and detailed exposed work, often by a team of skilled technicians.
While these sites have historically been difficult to date, with mixed assemblages accumulated over large time periods, advances in techniques such as uranium-series dating have allowed for greater accuracy. Historically, the excellent faunal record from East Africa has traditionally been used to compare sites based on relative dating. In this, the knowledge of environmental/faunal changes and extinction events allows us to know which hominin finds are relatively younger or older than others.
Research Highlight: The Taung Child
The well-known fossil of a juvenile Australopithecine, the “Taung Child,” was the first early hominin evidence ever discovered and was the first to demonstrate our common human heritage in Africa (Figure 9.15; Dart 1925). The tiny facial skeleton and natural endocast were discovered in 1924 by a local quarryman in the North West Province in South Africa and was painstakingly removed from the surrounding cement-like breccia by Raymond Dart using his wife’s knitting needles. When first shared with the scientific community in 1925, it was discounted as being nothing more than a young monkey of some kind. Prevailing biases of the time made it too difficult to contemplate that this small-brained hominin could have anything to do with our own history. The fact that it was discovered in Africa simply served to strengthen this bias.
It was not until adult specimens of the species began to be discovered at Sterkfontein Caves that scientific and public opinion began to be swayed. The most complete of these remains is best known as “Mrs. Ples” and was discovered in 1947 (Broom 1947).
The Taung Child is an excellent example of how understanding the morphology and physiology of teeth can reveal information about our evolution. While many introductory lectures will showcase how the foramen magnum is more anteriorly placed (indicating a head that sits centrally on the body, ideal for bipedalism), Taung Child also shows very human-like dentition. For one, the canine teeth were relatively small compared to other apes. In addition, there was little to no diastema (or gap) between the canines and incisors. The incisors themselves were vertical and close together, and not at an angle to the mouth (procumbent), as seen in other apes. Furthermore, the overall shape of the dental arcade was more rounded, or parabolic. Even though these features were related to deciduous teeth, they were overwhelmingly more human like than those seen in other apes.
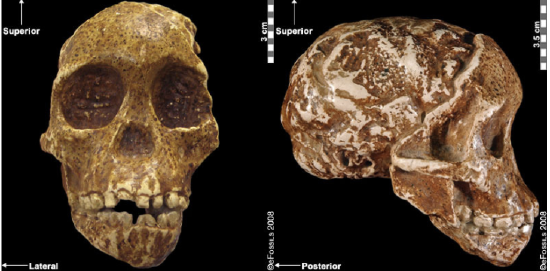 Figure \(\PageIndex{6}\): The Taung Child has a nearly complete face, mandible, and partial endocranial cast.
Figure \(\PageIndex{6}\): The Taung Child has a nearly complete face, mandible, and partial endocranial cast.The discovery of the Taung Child in 1924 shifted the focus of palaeoanthropological research from Europe to Africa although acceptance of this shift was slow (Dart 1925; Broom 1947). The species with which it is assigned, Australopithecus africanus (name meaning “Southern Ape of Africa”), is currently dated to between 3.3 mya and 2.1 mya (Pickering and Kramers 2010), with discoveries from Sterkfontein, Taung, Makapansgat, and Gladysvale in South Africa (Figure 9.16). A relatively large brain (400 cc to 500 cc), small canines without an associated diastema, and more rounded cranium and smaller teeth than Au. afarensis indicate some derived traits. Similarly, the postcranial remains (in particular, the pelvis) indicate bipedalism. However, the sloping face and curved phalanges (indicative of retained arboreal locomotor abilities) show some primitive features. Although not in direct association with stone tools, a 2015 study noted that the trabecular bone morphology of the hand was consistent with forceful tool manufacture and use, suggesting potential early tool abilities.
 Figure \(\PageIndex{7}\): An artistic reconstruction of Australopithecus africanus by John Gurche.
Figure \(\PageIndex{7}\): An artistic reconstruction of Australopithecus africanus by John Gurche.Another famous Au. africanus skull (the skull of “Mrs. Ples”) was previously attributed to Plesianthropus transvaalensis, meaning “near human from the Transvaal,” the old name for Gauteng Province, South Africa (Broom 1947; Broom 1950). The name was shortened by contemporary journalists to “Ples” (Figure 9.17). Due to the prevailing mores of the time, the assumed female found herself married, at least in name, and has become widely known as “Mrs. Ples.” It was later reassigned to Au. africanus and is now argued to be a young male rather than an adult female cranium (Thackeray 2000, Thackeray et al. 2002).
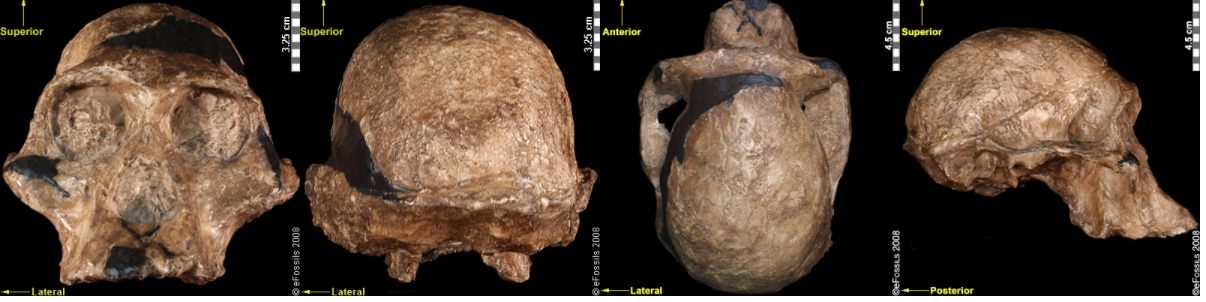 Figure \(\PageIndex{8}\): The “Mrs. Ples” brain case is small in size (like apes) but has a less prognathic face and its foramen magnum is positioned more like modern humans than in African apes.
Figure \(\PageIndex{8}\): The “Mrs. Ples” brain case is small in size (like apes) but has a less prognathic face and its foramen magnum is positioned more like modern humans than in African apes.In 2008, nine-year-old Matthew Berger, son of paleoanthropologist Lee Berger, noted a clavicle bone in some leftover mining breccia in the Malapa Fossil Site (South Africa). After rigorous studies, the species, Australopithecus sediba (meaning “fountain” or “wellspring” in the South African language of Sesotho), was named in 2010 (Figure 9.18; Berger et al. 2010). The first type specimen belongs to a juvenile male, Karabo (MH1), but the species is known from at least six partial skeletons of infants through adult. These specimens are currently dated to 1.97 mya (Dirks et al. 2010). The discoverers have argued that Au. sediba shows mosaic features between Au. africanus and Homo, which potentially indicates a transitional species, although this is heavily debated. These features include a small brain size (Australopithecus-like; 420 cc to 450 cc) but gracile mandible and small teeth (Homo-like). Similarly, the postcranial skeletons are also said to have mosaic features: scientists have interpreted this mixture of traits (such as a robust ankle but evidence for an arch in the foot) as a transitional phase between a body previously adapted to arborealism (tree climbing, particularly in evidence from the bones of the wrist) to one that adapted to bipedal ground walking. Some researchers have argued that Au. sediba shows a modern hand morphology (shorter fingers and a longer thumb), indicating that adaptations to tool manufacture and use may be present in this species.
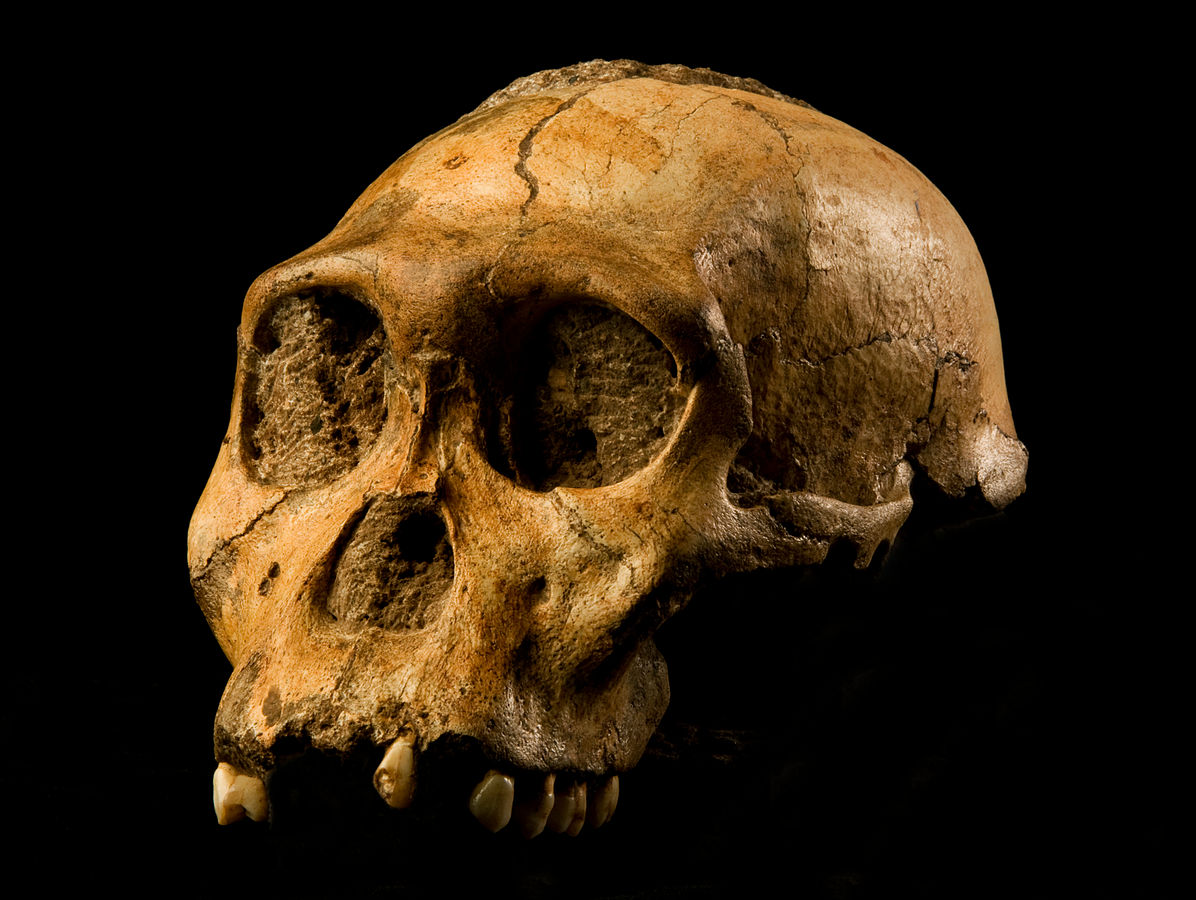 Figure \(\PageIndex{9}\): Australopithecus sediba shows mosaic features between Au. africanus and Homo.
Figure \(\PageIndex{9}\): Australopithecus sediba shows mosaic features between Au. africanus and Homo.Another famous Australopithecine find from South Africa is that of the nearly complete skeleton now known as “Little Foot” (Clarke 1998; Clarke 2013). Little Foot (StW 573) is potentially the earliest dated South African hominin fossil (3.7 mya, based on radiostopic techniques, although some argue that it is younger than 3 mya; Pickering and Kramers 2010). The name is jokingly in contrast to the cryptid species “bigfoot” and is named because the initial discovery of four ankle bones indicated bipedality. Little Foot was discovered by Ron Clarke in 1994, when he came across the ankle bones while sorting through monkey fossils in the University of Witwatersrand collections (Clarke and Tobias 1995). He asked Stephen Motsumi and Nkwane Molefe to identify the known records of the fossils, which allowed them to find the rest of the specimen within just days of searching the Sterkfontein Caves’ Silberberg Grotto. The discoverers of Little Foot insist that other fossil finds, previously identified as Au. Africanus, be placed in this new species based on shared primitive traits with older East African Australopithecines (Clarke and Kuman 2019). These include features such as a relatively large brain size (408 cc), robust zygomatic arch, and a flatter midface. Furthermore, the discoverers have argued that the heavy anterior dental wear patterns, relatively large anterior dentition, and smaller hind dentition of this specimen more closely resemble that of Au. anamensis or Au. afarensis.
It has thus been placed in the species Australopithecus prometheus. This species name refers to a previously defunct taxon named by Raymond Dart. The name “prometheus” refers to the Greek titan who stole fire from the gods to give to humanity. Raymond Dart believed that the bones he discovered at Makapansgat in South Africa reflected burning when it was later determined that they were in fact Au. africanus remains that had been stained by manganese during fossilization. The species designation was, through analyzing Little Foot, revived by Ron Clarke, who insists that many other fossil hominin specimens have prematurely been placed into Au. africanus. Others say that it is more likely that Au. africanus is a more variable species and not representative of two distinct species.
Paranthropus “Robust” Australopithecines
In the robust australopithecines, the specialized nature of the teeth and masticatory system, such as flaring zygomatic arches (cheekbone) to accommodate the large temporalis (chewing) muscle, indicated a shift in diet in these taxa. Some argued that the diet of the robust australopithecines was so specific that any change in environment would have accelerated their extinction. The generalist nature of the teeth of the gracile australopithecines, and certainly early Homo, would have made these hominins more capable of surviving through and adapting to to environmental change. However, some have suggested that the features of the robust australopithecines might have developed more in response to effectively eating fallback foods in hard times rather than indicating a lack of adaptability.
Paranthropus is usually referred to by scholars as the “robust” australopithecine, because of its defining distinct morphological features. Features that are closer to those of the assumed ancestral type are referred to as P. aethiopicus, and those that have become derived are referred to as both P. boisei and P. robustus (Strait et al. 1997; Wood and Schroer 2017). These features include a large, broad, dish-shaped face and zygomatic arches that are forward facing, including a large mandible with extremely large posterior dentition. These three species have been grouped together by a majority of scholars as a genus as they share more derived features (are more closely related to each other; or, in other words, are monophyletic) than the other australopithecines (Wood 2010; Hlazo 2015; Grine 1988; Strait et al. 1997). Much of the phylogenetic debate (and research, more generally) has revolved around the shared adaptations of these “robust” australopithecines linked to a diet of hard and/or tough foods (Brain 1967; Rak 1988). This includes their large posterior dentition (they are referred to as megadonts), hyper-thick enamel, thick robust jaws, and flared zygomatic arches (Kimbel 2015; Lee-Thorp 2011; Wood 2010).
In terms of diet, the tougher, chewing diets of the robust australopithecines are supported by the extreme morphology of their face and cranium. Similarly, the prognathic faces, which jut out under the eyes in the earlier hominins, are less pronounced than in those of living great apes, yet are more pronounced than in early Homo. In contrast, the orthognathic (flat) face of our genus is well suited to accommodate our relatively small generalized teeth and processed diets.
Researchers have mostly reached a consensus to the umbrella term Paranthropus. However, there are those who remain cautious/disagree (Constantino and Wood 2004; Constantino and Wood 2007; Wood 2010). As a collective, this genus spans 2.7 mya to 1.0 mya, although the dates of the individual species differ. The genus was first discovered in Kromdraai B, South Africa, by Robert Broom, who first attributed the holotype of specimen TM 1517 (Broom 1938a; Broom 1938b; Broom 1950; Hlazo 2018).
The earliest of the Paranthropus species, Paranthropus aethiopicus, is dated to between 2.7 mya and 2.3 mya and is currently found in Tanzania, Kenya, and Ethiopia in the EARS system (Figure 9.19; Walker et al. 1986; Constantino and Wood 2007; Hlazo 2015; Kimbel 2015; White 1988). It is well known because of the “Black Skull” (KNM–WT 17000), so called because of the mineral manganese that stained it black during fossilization (Kimbel 2015). As with all robust Australopithecines, P. aethiopicus has the shared derived traits of large, flat premolars and molars; large, flaring zygomatic arches for accommodating large chewing muscles (the temporalis muscle); a sagittal crest for increased muscle attachment of the chewing muscles to the skull; and a robust mandible and supraorbital torus (brow ridge). However, only a few teeth have been found. A proximal tibia indicates bipedality and similar body size to Au. afarensis. In recent years, researchers have discovered and assigned a proximal tibia and juvenile cranium (L.338y-6) to the species (Wood and Boyle 2016).
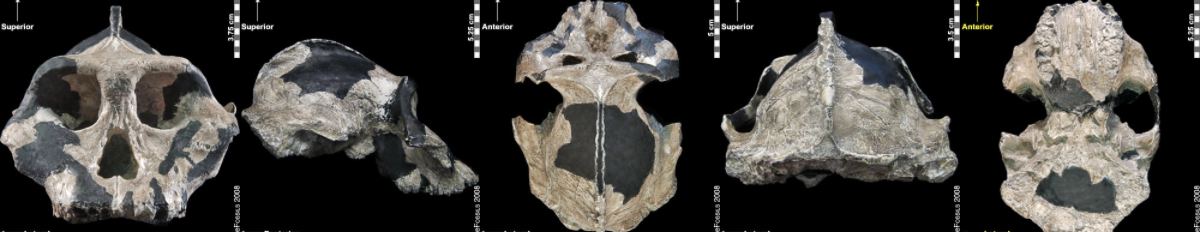 Figure \(\PageIndex{10}\): The “Black Skull” (Paranthropus aethiopicus) had a large sagittal crest and large, flared zygomatic arches that indicate it had large chewing muscles and a powerful biting force.
Figure \(\PageIndex{10}\): The “Black Skull” (Paranthropus aethiopicus) had a large sagittal crest and large, flared zygomatic arches that indicate it had large chewing muscles and a powerful biting force. Figure \(\PageIndex{11}\): Artistic reconstruction of a Paranthropus boisei, male, by John Gurche.
Figure \(\PageIndex{11}\): Artistic reconstruction of a Paranthropus boisei, male, by John Gurche.First attributed as Zinjanthropus boisei (with the first discovery going by the nickname “Zinj” or sometimes “Nutcracker Man”), Paranthropus boisei was discovered in 1959 by Mary Leakey (see Figure 9.20 and 9.21; Hay 1990; Leakey 1959). This “robust” australopith species is distributed across countries in East Africa at sites such as Kenya (Koobi Fora, West Turkana, and Chesowanja), Malawi (Malema-Chiwondo), Tanzania (Olduvai Gorge and Peninj), and Ethiopia (Omo River Basin and Konso). The hypodigm has been found by researchers to date to roughly 2.4 mya to 1.4 mya. Due to the nature of its exaggerated, larger, and more robust features, P. boisei has been termed —that is, even more heavily built than other robust forms, with very large, flat posterior dentition (largest of all hominins currently known; Kimbel 2015). Richard Leakey and Bernard Wood have both suggested that P. boisei could have made and used stone tools. Tools dated to 2.5 mya in Ethiopia have been argued to possibly belong to this species. Despite the cranial features of P. boisei indicating a tough diet of tubers, nuts, and seeds, isotopes indicate a diet high in C4 foods (e.g., grasses, such as sedges). This differs from what is seen in P. robustus. Another famous specimen from this species is the Peninj mandible from Tanzania, found in 1964 by Kimoya Kimeu.
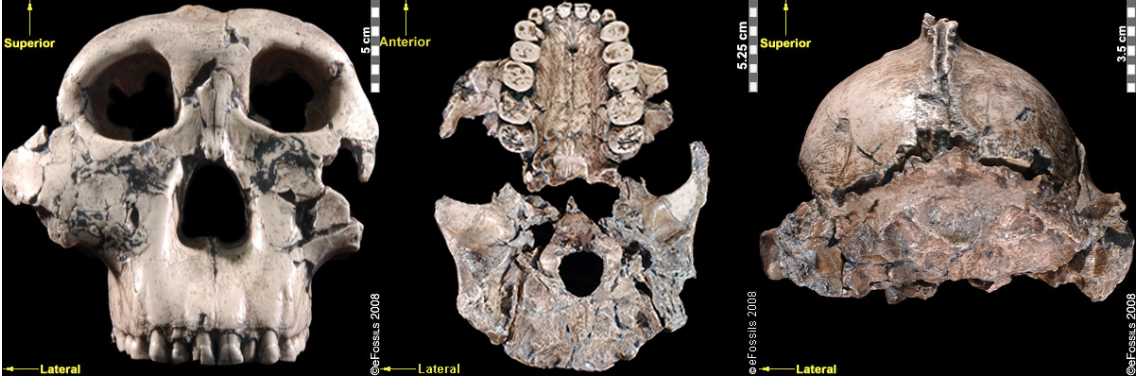 Figure \(\PageIndex{12}\): “Nutcracker Man” (Paranthropus boisei) had hyper-robust features including very large dentition, flaring zygomatic arches, a broad concave face. It had a powerful and extremely efficient chewing force.
Figure \(\PageIndex{12}\): “Nutcracker Man” (Paranthropus boisei) had hyper-robust features including very large dentition, flaring zygomatic arches, a broad concave face. It had a powerful and extremely efficient chewing force.Paranthropus robustus was the first taxon to be discovered within the genus in Kromdraai B by a school boy named Gert Terblanche, and subsequent fossil discoveries were made by researcher Robert Broom in 1938 (Figure 9.22; Broom, 1938a; Broom 1938b; Broom 1950). Paranthropus robustus dates approximately to 2.0 mya to 1 mya and is the only taxon from the genus to be discovered in South Africa. It has been found in sites all over the Cradle, such as Kromdraai B, Swartkrans, Gondolin, Drimolen, and Coopers Cave. Several of these fossils are fragmentary in nature, distorted, and not well preserved because they have been recovered from quarry breccia using explosives. P. robustus features are neither as “hyper-robust” as P. boisei nor as primitive as P. aethiopicus; instead, they have been described as being less derived, more general features that are shared with both East African species (e.g., the sagittal crest and zygomatic flaring) (Rak 1983; Walker and Leakey 1988). Enamel hypoplasia is also common in this species, possibly because of instability in the development of large, thick-enameled dentition.
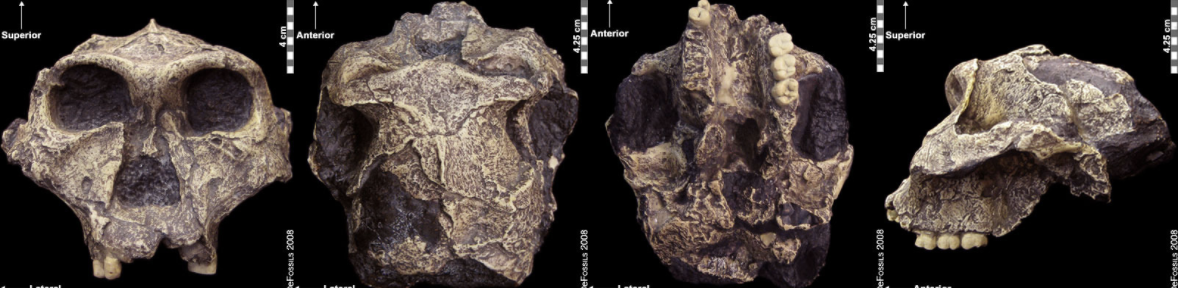 Figure \(\PageIndex{13}\): Paranthropus robustus had less derived, more general features; it was not as robust as P. boisei and not as primitive as P. aethiopicus. It also appeared to be prone to a tooth defect (enamel hypoplasia), making the enamel thinner and more prone to decay.
Figure \(\PageIndex{13}\): Paranthropus robustus had less derived, more general features; it was not as robust as P. boisei and not as primitive as P. aethiopicus. It also appeared to be prone to a tooth defect (enamel hypoplasia), making the enamel thinner and more prone to decay.Comparisons between Gracile and Robust Australopiths
Comparisons between gracile and robust australopithecines may indicate different phylogenetic groupings but may also indicate parallel evolution in several species. In general, the robust australopithecines have large temporalis (chewing) muscles, as indicated by flaring zygomatic arches, sagittal crests, and robust mandibles (jawbones). Their hind dentition is large (megadont), with low cusps and thick enamel. Within the gracile australopithecines, researchers have debated the relatedness of the species, or even whether these species should be lumped together to represent more variable or polytypic species. Often researchers will attempt to draw chronospecific trajectories, with one taxon evolving into another over time.

