14.1: Overview of Brain Mechanisms in Intelligence, Language, and Cognition
- Page ID
- 113221
This page is a draft and under active development. Please forward any questions, comments, and/or feedback to the ASCCC OERI (oeri@asccc.org).
- Discuss the general relationship across species between brain size, body size, and intelligence, including trends in corticalization and cortical folding
- Describe the functions of glial cells
- Describe the locations and functions of each of the lobes of the cerebral cortex
- Describe contralateral control
- Discuss general principles that determine the amounts of somatosensory and motor cortex devoted to each part of the body
- Describe visual agnosia and the location of the brain damage that causes it
- Describe neuroplasticity and briefly discuss examples of it
- Discuss what the split-brain experiments tell us about cerebral lateralization of function
Overview
We begin this chapter with a general review of the brain areas involved in complex psychological functions, focusing on the cerebral cortex, including studies of the split brain. Although an introductory psychology course is a prerequisite for this course in Biological Psychology, a refresher will be useful for you, the student, in preparation for the detail to be discussed in subsequent modules of this chapter.
The Cerebral Cortex Creates Intelligence, Language, and Thinking: Overview of the Basics
All animals have adapted to their environments by developing abilities that help them survive. Some animals have hard shells, others run extremely fast, and some have acute hearing. Human beings do not have any of these particular characteristics, but we do have one big advantage over other animals—we are very, very smart.
You might think that we should be able to determine the intelligence of an animal by looking at the ratio of the animal’s brain weight to the weight of its entire body. But this does not really work. The elephant’s brain is one thousandth of its weight, but the whale’s brain is only one ten-thousandth of its body weight. On the other hand, although the human brain is one 60th of its body weight, the mouse’s brain represents one fortieth of its body weight. Despite these comparisons, elephants do not seem 10 times smarter than whales, and humans definitely seem smarter than mice.
The key to the advanced intelligence of humans is not found in the size of our brains. What sets humans apart from other animals is our larger cerebral cortex—the outer bark-like layer of our brain that allows us to so successfully use language, acquire complex skills, create tools, and live in social groups (Gibson, 2002). In humans, the cerebral cortex is wrinkled and folded, rather than smooth as it is in most other animals. This creates a much greater surface area and size, and allows increased capacities for learning, remembering, and thinking. The increasing amount of cortex in mammals involving folding of the cerebral cortex is referred to as corticalization.
Although the cortex is only about one tenth of an inch thick, it makes up more than 80% of the brain’s weight. The human cortex contains about 20 billion nerve cells (the entire human brain has somewhere between 100 and 200 billion neurons) and at least 300 trillion synaptic connections (de Courten-Myers, 1999). Supporting all these neurons are billions more glial cells (glia), cells that surround and link to the neurons, protecting them, providing them with nutrients, and absorbing unused neurotransmitters. The glia come in different forms and have different functions. For instance, the myelin sheath surrounding the axon of many neurons is a type of glial cell. The glia are essential partners of neurons, without which the neurons could not survive or function (Miller, 2005).
As you recall from the chapter on anatomy of the nervous system, the cerebral cortex is divided into two hemispheres, and each hemisphere is divided into four lobes, each separated by little "valleys" known as fissures (the little hills between fissures are called gyri, singular, gyrus).
If we look at the cortex starting at the front of the brain and moving over the top (see Figure 14.1.1), we see first the frontal lobe (behind the forehead), which is responsible primarily for thinking, planning, memory, and judgment. In addition, in most people the left frontal lobe contains Broca's area essential for speech and other language functions (in some left handers, Broca's area may be in the right frontal lobe or language may be less lateralized and instead is spread more equally between the two hemispheres; thus, left handers tend to lose less language function than right handers if they suffer damage to left frontal lobe).
Following the frontal lobe is the parietal lobe, which extends from the middle to the back of the skull and which is responsible primarily for processing information about touch and spatial perception, and parts of which appear to be involved in visualization (discussed later in this chapter). Spatial perception is one component of intelligence measured on IQ tests. Damage to parts of the right parietal association cortex produces unilateral neglect, the inability to understand that the left side of your body belongs to you--thus it is "neglected" to the point where a patient may not be able "to find" their left arm, or may dress and groom only the right side of their body.
Then comes the occipital lobe, at the very back of the skull, which processes visual information (the central region of the occipital lobe is called primary visual cortex and has a point-for-point mapping of the retina on its surface; surrounding the primary visual cortex is visual association cortex which is involved in higher order, more complex visual processing and perception, and along with primary visual cortex appears to store long-term visual memories).
Finally, in front of the occipital lobe (near the temples and ears) is the temporal lobe, responsible primarily for hearing and language (Wernicke's Area is here in the left temporal lobe in most people and interacts with Broca's area in the left Frontal lobe in language processing). Also located in the temporal lobe is the Inferotemporal (IT) cortex which is involved in visual recognition. Damage in IT cortex produces visual agnosia--people can see and describe visual detail but they can't put the details together to recognize what it is that they are looking at. A small area of the right inferior temporal cortex known as the fusiform face area (FFA) appears to be specialized for face processing (Kanwisher & Yovel, 2006). Damage to the FFA produces a related, but more specific disorder, prosopagnosia or face blindness, the inability to recognize familiar faces, even those of close family members, and even one's own face in photographs (see Barton et al., 2002).
Another area involved in facial recognition in humans is the occipital face area (OFA) located in the lateral occipital lobe near the occipital gyrus. Brain damage in the OFA results in impaired face recognition. Research suggests that there may be a topographic face map in the OFA where adjacent areas of the human face are represented by adjacent areas of cortex in the OFA (Henriksson, et al., 2015). The FFA and OFA are interconnected and are part of a network for face processing and face recognition in the human inferior temporal and lateral occipital cortices.
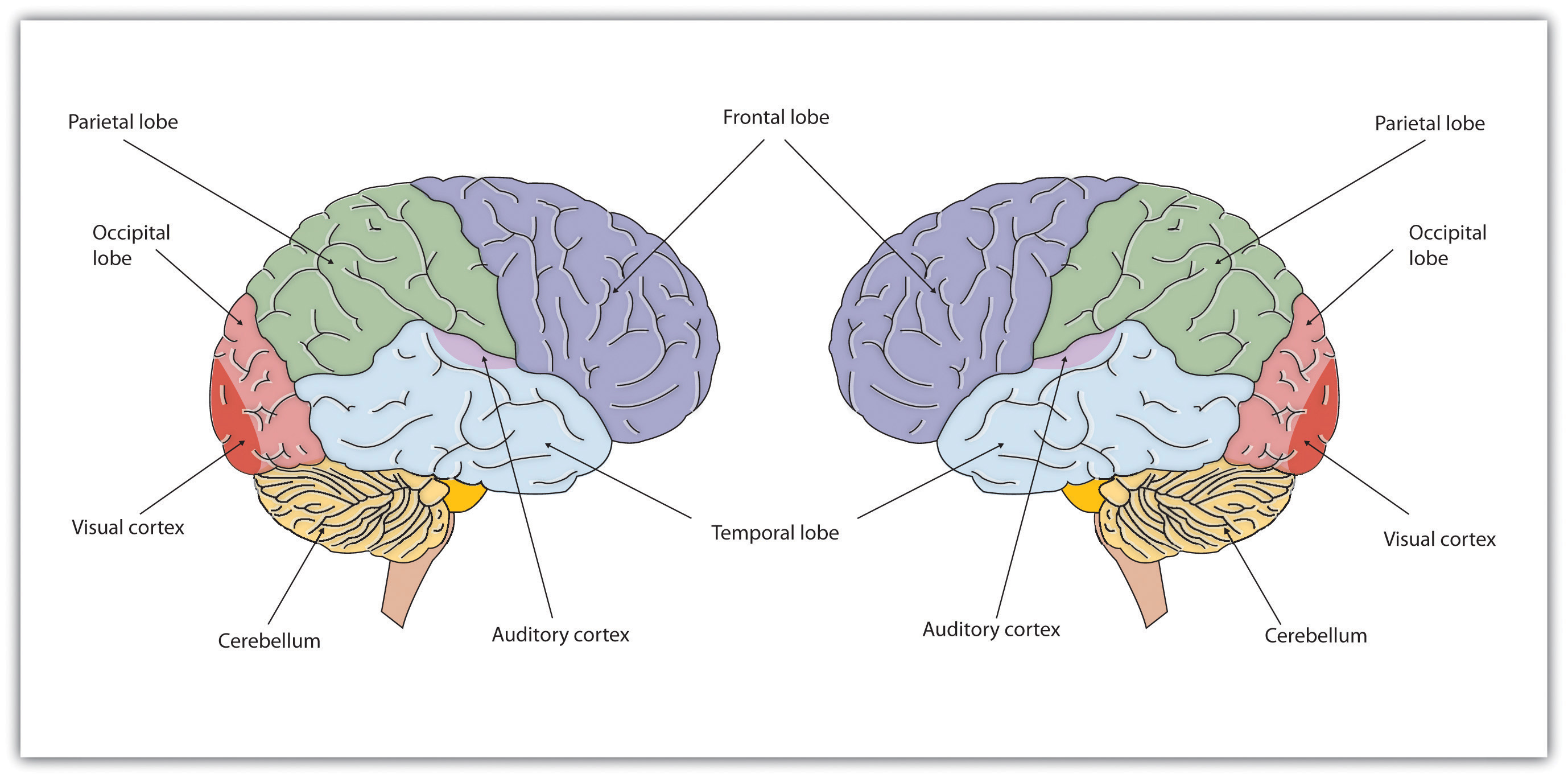
Figure \(\PageIndex{1}\): The brain is divided into two hemispheres (right and left), each of which has four lobes (temporal, frontal, occipital, and parietal). Furthermore, there are specific cortical areas within the lobes that control different processes. The FFA in interior temporal lobe and the OFA in the occipital lobe are specialized for facial processing. Specialized areas of cortex for specific functions illustrate the principle of localization of function within the cerebral cortex.
Functions of the Cortex
When the German physicists Gustav Fritsch and Eduard Hitzig (1870/2009) applied mild electric stimulation to different parts of a dog’s cortex, they discovered that they could make different parts of the dog’s body move. Furthermore, they discovered an important and unexpected principle of brain activity. They found that stimulating the right side of the brain produced movement in the left side of the dog’s body, and vice versa. This finding follows from a general principle about how the brain is structured, called contralateral control. The brain is wired such that in most cases the left hemisphere receives sensations from and controls the right side of the body, and vice versa.
Fritsch and Hitzig also found that the movement that followed the brain stimulation only occurred when they stimulated a specific arch-shaped region that runs across the top of the brain from ear to ear, just at the front of the parietal lobe (see Figure 14.1.2 below). Fritsch and Hitzig had discovered the motor cortex, the part of the cortex that controls and executes movements of the body by sending signals to the cerebellum and the primary motor neurons of the spinal cord. More recent research has mapped the motor cortex even more fully, by applying mild electronic stimulation to different areas of the motor cortex in conscious patients while observing their bodily responses (because the brain has no sensory receptors, these patients feel no pain). As you can see in Figure 14.1.2, this research has revealed that the motor cortex is specialized for providing control over the body; there is a topographic representation of the body surface on the surface of the motor cortex. In fact, the parts of the body that require more precise and finer movements, such as the face and the hands, are allotted the largest areas of motor cortical space. This makes sense since finer movements require more processing and thus more cortical tissue.
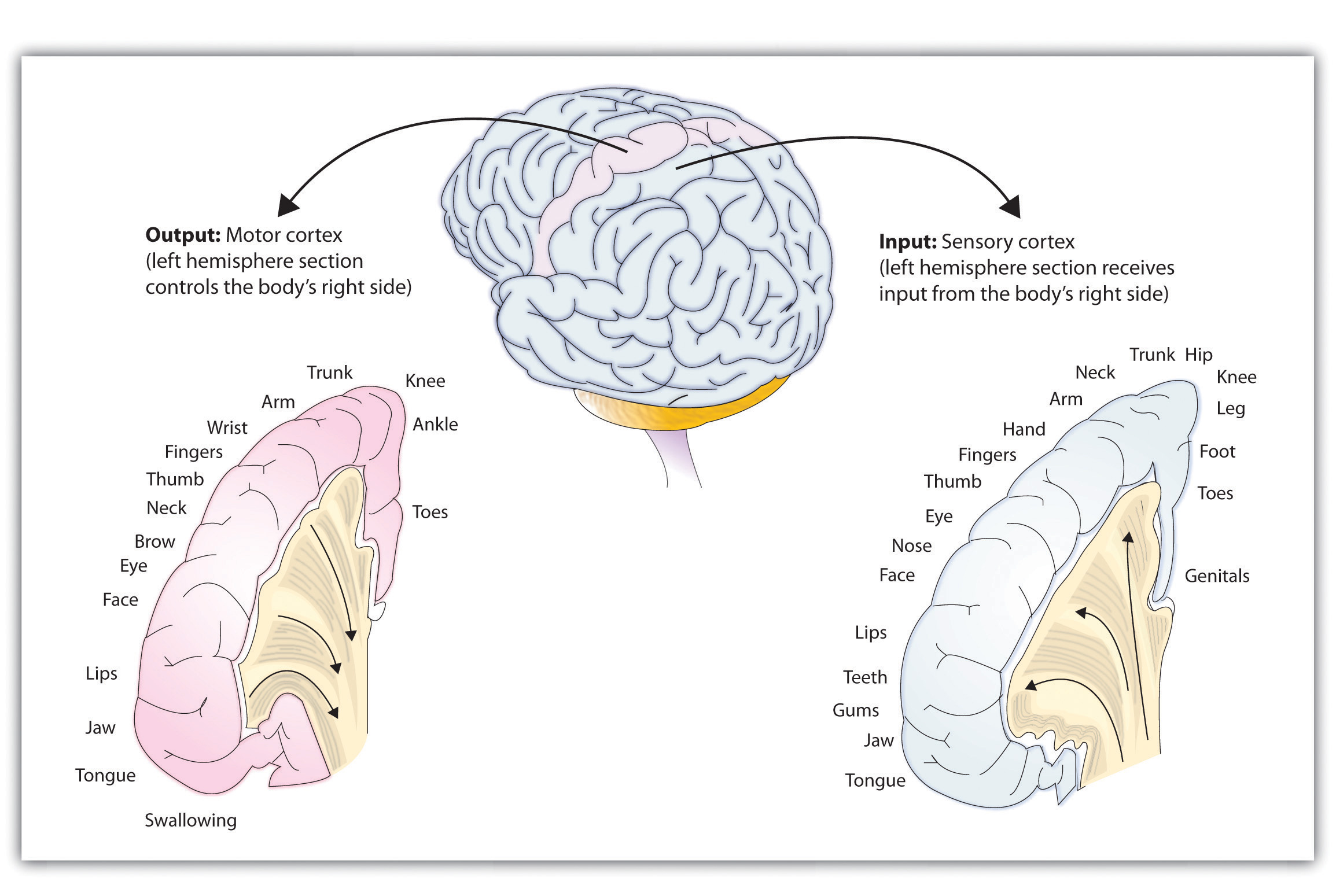
Figure \(\PageIndex{2}\): The Somatosensory Cortex and the Motor Cortex.
The portion of the sensory and motor cortex devoted to receiving messages that control specific regions of the body is determined by the amount of fine movement that area is capable of performing. Thus the hand and fingers have as much area in the cerebral cortex as does the entire trunk of the body.
Just as the primary motor cortex (the precentral gyrus, at the back of the frontal lobe) sends out messages to the specific parts of the body, the primary somatosensory cortex (the post-central gyrus, just behind the central fissure), an area just behind and parallel to the motor cortex, receives information from the skin’s sensory receptors and the movements of different body parts. Again, the more sensitive the body region, the more area is dedicated to it in the sensory cortex. Our sensitive lips, for example, occupy a large area in the sensory cortex, as do our fingers and genitals, whereas the trunk of the body has relatively less area in the sensory cortex devoted to it.
Other areas of the cortex process other types of sensory information. The visual cortex is the area located in the occipital lobe (at the very back of the brain) that processes visual information. If your brain were stimulated, while conscious on the operating table, by an electrode in the visual cortex, you would see flashes of light or color. Perhaps you remember having had the experience of “seeing stars” when you were hit or fell on the back of your head (this finding suggests that the luminosity and color that we attribute to light are really produced in the brain, when occipital cortex is activated, and that these sensations don't exist in the exterior world outside our heads--luminosity and color are not properties of the world, but are just brain/psychological codes for dark and colorless electromagnetic energies of various wavelengths in the visible spectrum which only become luminous and colored after they are transduced into neuron potentials which activate visual system neurons). The temporal lobe, located on the lower side of each hemisphere, near your temples, which contains auditory cortex as well as Wernicke's area (on the left temporal lobe of most people) responsible for hearing and language comprehension, respectively. The inferior temporal lobe (IT cortex) also processes some visual information, providing us, as discussed above, with the ability to recognize and name the objects around us (Martin, 2007).
As you can see in Figure 14.1.2, the motor and sensory areas of the cortex account for a relatively small part of the total cortex. The remainder of the cortex is made up of association areas of cortex in which sensory and motor information is combined and associated with our stored knowledge. These association areas of cortex (to be discussed in more detail later in this chapter) are the places in the brain that are responsible for most of the things that make human beings seem human. The association areas are involved in higher mental functions, such as learning, thinking, planning, judging, moral reflecting, figuring, and spatial reasoning.
The Brain Is Flexible: Neuroplasticity
The control of some specific bodily functions, such as movement, vision, and hearing, is performed in specified areas of the cortex, and if these areas are damaged, the individual will likely lose the ability to perform the corresponding function. For instance, if an infant suffers damage to facial recognition areas in the temporal lobe (FFA), it is likely that he or she will never be able to recognize faces (Farah, Rabinowitz, Quinn, & Liu, 2000). On the other hand, the brain is not divided up in an entirely rigid way. The brain’s neurons have a remarkable capacity to reorganize and extend themselves to carry out particular functions in response to the needs of the organism, and to repair damage. As a result, the brain constantly creates new neural communication routes and rewires existing ones. Neuroplasticity refers to the brain’s ability to change its structure and function in response to experience or damage. Neuroplasticity enables us to learn and remember new things and adjust to new experiences as discussed in chapter 10 on learning and memory.
Our brains are the most “plastic” when we are young children, as it is during this time that we learn the most about our environment. On the other hand, neuroplasticity continues to be observed even in adults (Kolb & Fantie, 1989). The principles of neuroplasticity help us understand how our brains develop to reflect our experiences. For instance, accomplished musicians have a larger auditory cortex compared with the general population (Bengtsson et al., 2005) and also require less neural activity to move their fingers over the keys than do novices (Münte, Altenmüller, & Jäncke, 2002). This is because accomplished musicians through practice have eliminated synaptic connections associated with incorrect movements, making neural control more efficient (see chapter 10 on long-term depression. LTD).
Plasticity is also observed when there is damage to the brain or to parts of the body that are represented in the motor and sensory cortexes. When a tumor in the left hemisphere of the brain impairs language, the right hemisphere will begin to compensate to help the person recover the ability to speak (Thiel et al., 2006). And if a person loses a finger, the area of the sensory cortex that previously received information from the missing finger will begin to receive input from adjacent fingers, causing the remaining digits to become more sensitive to touch (Fox, 1984).
Although neurons cannot repair or regenerate themselves as skin or blood vessels can, new evidence suggests that the brain can engage in neurogenesis, the forming of new neurons (Van Praag, Zhao, Gage, & Gazzaniga, 2004). These new neurons originate deep in the brain and may then migrate to other brain areas where they form new connections with other neurons (Gould, 2007). This leaves open the possibility that someday scientists might be able to “rebuild” damaged brains by creating drugs that help grow neurons.
Research Focus: Identifying the Unique Functions of the Left and Right Hemispheres Using Split-Brain Patients
We have seen that the left hemisphere of the brain primarily senses and controls the motor movements on the right side of the body, and vice versa. This fact provides an interesting way to study brain lateralization—the idea that the left and the right hemispheres of the brain are specialized to perform different functions. Gazzaniga, Bogen, and Sperry (1965) studied a patient, known as W. J., who had undergone an operation to relieve severe seizures. In this surgery the region that normally connects the two halves of the brain and supports communication between the hemispheres, known as the corpus callosum, is severed. As a result, the patient essentially becomes a person with two separate brains. Because the left and right hemispheres are separated, each hemisphere develops a mind of its own, with its own sensations, concepts, and motivations (Gazzaniga, 2005).
In their research, Gazzaniga and his colleagues tested the ability of W. J. to recognize and respond to objects and written passages that were presented to only the left or to only the right brain hemispheres (see Figure 14.1.3). The researchers had W. J. look straight ahead and then flashed, for a fraction of a second, a picture of a geometrical shape to the left of where he was looking. By doing so, they assured that—because the two hemispheres had been separated—the image of the shape was experienced only in the right brain hemisphere (remember that sensory input from the left side of the body is sent to the right side of the brain). Gazzaniga and his colleagues found that W. J. was able to identify what he had been shown when he was asked to pick the object from a series of shapes, using his left hand, but that he could not do this when the object was shown in the right visual field. On the other hand, W. J. could easily read written material presented in the right visual field (and thus experienced in the left hemisphere) but not when it was presented in the left visual field.
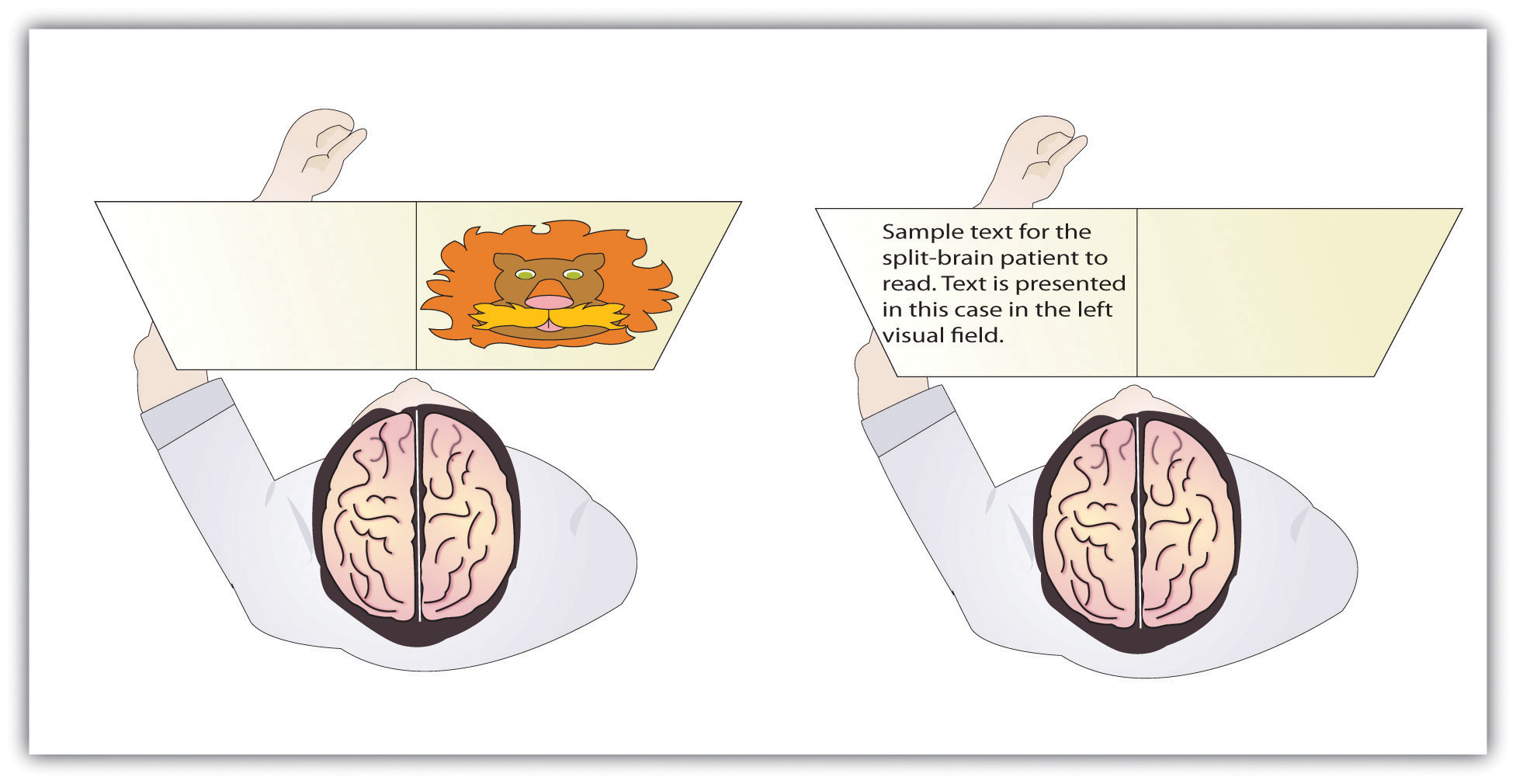
Figure \(\PageIndex{3}\): Visual and Verbal Processing in the Split-Brain Patient
The information that is presented on the left side of our field of vision is transmitted to the right brain hemisphere, and vice versa. In split-brain patients, the severed corpus callosum does not permit information to be transferred between hemispheres (as it is in normal persons without brain damage), which allows researchers to learn about the functions of each hemisphere. In the perceptual test on the left, the split-brain patient could not choose which image had been previously presented because the left hemisphere cannot process visual information. In the test on the right side of Figure 14.1.3, the patient could not read the passage because the right brain hemisphere cannot process language.
This research, and many other studies following it, has demonstrated that the two brain hemispheres specialize in different abilities. In most people the ability to speak, write, and understand language is located in the left hemisphere. This is why W. J. could read passages that were presented on the right side and thus transmitted to the left hemisphere, but could not read passages that were only experienced in the right brain hemisphere. The left hemisphere is also better at math and at judging time and rhythm. It is also superior in coordinating the order of complex movements—for example, lip movements needed for speech. The right hemisphere, on the other hand, has only very limited verbal abilities, and yet it excels in perceptual skills. The right hemisphere is able to recognize objects, including faces, patterns, and melodies, and it can put a puzzle together or draw a picture. This is why W. J. could pick out the image when he saw it on the left, but not the right, visual field.
Although Gazzaniga’s research demonstrated that the brain is in fact lateralized, such that the two hemispheres specialize in different activities, this does not mean that when people behave in a certain way or perform a certain activity they are only using one hemisphere of their brains at a time. That would be drastically oversimplifying the concept of brain differences. We normally use both hemispheres at the same time, and the difference between the abilities of the two hemispheres is not absolute (Soroker et al., 2005).
Why Are Some People Left-Handed?
Across cultures and ethnic groups, about 90% of people are mainly right-handed, whereas only 10% are primarily left-handed (Peters, Reimers, & Manning, 2006). This fact is puzzling, in part because the number of left-handers is so low, and in part because other animals, including our closest primate relatives, do not show any type of handedness. The existence of right-handers and left-handers provides an interesting example of the relationship among evolution, biology, and social factors and how the same phenomenon can be understood at different levels of analysis (Harris, 1990; McManus, 2002).
At least some handedness is determined by genetics. Ultrasound scans show that 9 out of 10 fetuses suck the thumb of their right hand, suggesting that the preference is determined before birth (Hepper, Wells, & Lynch, 2005), and the mechanism of transmission has been linked to a gene on the X chromosome (Jones & Martin, 2000). It has also been observed that left-handed people are likely to have fewer children, and this may be in part because the mothers of left-handers are more prone to miscarriages and other prenatal problems (McKeever, Cerone, Suter, & Wu, 2000).
But culture also plays a role. In the past, left-handed children were forced to write with their right hands in many countries, and this practice continues, particularly in collectivistic cultures, such as India and Japan, where left-handedness is viewed negatively as compared with individualistic societies, such as the United States. For example, India has about half as many left-handers as the United States (Ida & Mandal, 2003).
There are both advantages and disadvantages to being left-handed in a world where most people are right-handed. One problem for lefties is that the world is designed for right-handers. Automatic teller machines (ATMs), classroom desks, scissors, microscopes, drill presses, and table saws are just some examples of everyday machinery that is designed with the most important controls on the right side. This may explain in part why left-handers suffer somewhat more accidents than do right-handers (Dutta & Mandal, 2006).
Despite the potential difficulty living and working in a world designed for right-handers, there seem to be some advantages to being left-handed. Throughout history, a number of prominent artists have been left-handed, including Leonardo da Vinci, Michelangelo, Pablo Picasso, and Max Escher. Because the right hemisphere is superior in imaging and visual abilities, there may be some advantage to using the left hand for drawing or painting (Springer & Deutsch, 1998). Left-handed people are also better at envisioning three-dimensional objects, which may explain why there is such a high number of left-handed architects, artists, and chess players in proportion to their numbers (Coren, 1992). However, there are also more left-handers among those with reading disabilities, allergies, and migraine headaches (Geschwind & Behan, 2007), perhaps due to the fact that a small minority of left-handers owe their handedness to a birth trauma, such as being born prematurely (Betancur, Vélez, Cabanieu, & le Moal, 1990). Interestingly, there have been a disproportionally large number of U.S. Presidents who have been left-handed, including Gerald Ford, George H.W. Bush, Bill Clinton, and Barack Obama.
Summary
The evolutionarily old brain—including the brain stem, medulla, pons, reticular formation, thalamus, cerebellum, amygdala, hypothalamus, and hippocampus—regulates basic survival functions, such as breathing, moving, resting, feeding, emotions, and memory.
The cerebral cortex, made up of billions of neurons and glial cells, is divided into the right and left hemispheres and into four lobes. The frontal lobe is primarily responsible for thinking, planning, memory, and judgment. The parietal lobe is primarily responsible for bodily sensations and touch. The temporal lobe is primarily responsible for hearing and language. The occipital lobe is primarily responsible for vision. Other areas of the cortex act as association areas, responsible for integrating information. The motor cortex controls voluntary movements. Body parts requiring the most control and dexterity take up the most space in the motor cortex. The sensory cortex receives and processes bodily sensations. Body parts that are the most sensitive occupy the greatest amount of space in the sensory cortex.
The brain changes as a function of experience and potential damage in a process known as plasticity. Neuroplasticity allows the brain to adapt and change as a function of experience or damage. The brain can generate new neurons through neurogenesis.
The severing of the corpus callosum, which connects the two hemispheres, creates a “split-brain patient,” with the effect of creating two separate minds operating in one person. Studies with split-brain patients as research participants have been used to study brain lateralization. The left cerebral hemisphere is primarily responsible for language and speech in most people, whereas the right hemisphere specializes in spatial and perceptual skills, visualization, and the recognition of patterns, faces, and melodies.
References
Barton, J. J., Press, D. Z., Keenan, J. P., & O’Connor, M. (2002). Lesions of the fusiform face area impair perception of facial configuration in prosopagnosia. Neurology, 58 (1), 71-78.
Bengtsson, S. L., Nagy, Z., Skare, S., Forsman, L., Forssberg, H., & Ullén, F. (2005). Extensive piano practicing has regionally specific effects on white matter development. Nature Neuroscience, 8 (9), 1148–1150.
Betancur, C., Vélez, A., Cabanieu, G., & le Moal, M. (1990). Association between left-handedness and allergy: A reappraisal. Neuropsychologia, 28(2), 223–227.
Bodmer, W., & McKie, R. (1994). The book of man: The quest to discover our genetic heritage. London, England: Little, Brown and Company.
Coren, S. (1992). The left-hander syndrome: The causes and consequences of left-handedness. New York, NY: Free Press.
de Courten-Myers, G. M. (1999). The human cerebral cortex: Gender differences in structure and function. Journal of Neuropathology and Experimental Neurology, 58, 217–226.
Dutta, T., & Mandal, M. K. (2006). Hand preference and accidents in India. Laterality: Asymmetries of Body, Brain, and Cognition, 11, 368–372.
Farah, M. J., Rabinowitz, C., Quinn, G. E., & Liu, G. T. (2000). Early commitment of neural substrates for face recognition. Cognitive Neuropsychology, 17(1–3), 117–123.
Fox, J. L. (1984). The brain’s dynamic way of keeping in touch. Science, 225(4664), 820–821.
Fritsch, G., & Hitzig, E. (2009). Electric excitability of the cerebrum (Über die Elektrische erregbarkeit des Grosshirns). Epilepsy & Behavior, 15(2), 123–130. (Original work published 1870)
Gazzaniga, M. S., Bogen, J. E., & Sperry, R. W. (1965). Observations on visual perception after disconnexion of the cerebral hemispheres in man. Brain, 88(2), 221–236.
Gazzaniga, M. S. (2005). Forty-five years of split-brain research and still going strong. Nature Reviews Neuroscience, 6(8), 653–659.
Geschwind, N., & Behan, P. (2007). Left-handedness: Association with immune disease, migraine, and developmental learning disorder. Cambridge, MA: MIT Press.
Gibson, K. R. (2002). Evolution of human intelligence: The roles of brain size and mental construction. Brain Behavior and Evolution 59, 10–20.
Gould, E. (2007). How widespread is adult neurogenesis in mammals? Nature Reviews Neuroscience 8, 481–488. doi:10.1038/nrn2147
Harris, L. J. (1990). Cultural influences on handedness: Historical and contemporary theory and evidence. In S. Coren (Ed.), Left-handedness: Behavioral implications and anomalies. New York, NY: Elsevier.
Henriksson, L., Mur, M., & Kriegeskorte, N. (2015). Faciotopy—A face-feature map with face-like topology in the human occipital face area. Cortex, 72, 156-167.
Hepper, P. G., Wells, D. L., & Lynch, C. (2005). Prenatal thumb sucking is related to postnatal handedness. Neuropsychologia, 43, 313–315.
Ida, Y., & Mandal, M. K. (2003). Cultural differences in side bias: Evidence from Japan and India. Laterality: Asymmetries of Body, Brain, and Cognition, 8(2), 121–133.
Jones, G. V., & Martin, M. (2000). A note on Corballis (1997) and the genetics and evolution of handedness: Developing a unified distributional model from the sex-chromosomes gene hypothesis. Psychological Review, 107(1), 213–218.
Kanwisher, N., & Yovel, G. (2006). The fusiform face area: a cortical region specialized for the perception of faces. Philosophical Transactions of the Royal Society B: Biological Sciences, 361(1476), 2109-2128.
Kolb, B., & Fantie, B. (1989). Development of the child’s brain and behavior. In C. R. Reynolds & E. Fletcher-Janzen (Eds.), Handbook of clinical child neuropsychology (pp. 17–39). New York, NY: Plenum Press.
Martin, A. (2007). The representation of object concepts in the brain. Annual Review of Psychology, 58, 25–45.
McKeever, W. F., Cerone, L. J., Suter, P. J., & Wu, S. M. (2000). Family size, miscarriage-proneness, and handedness: Tests of hypotheses of the developmental instability theory of handedness. Laterality: Asymmetries of Body, Brain, and Cognition, 5(2), 111–120.
McManus, I. C. (2002). Right hand, left hand: The origins of asymmetry in brains, bodies, atoms, and cultures. Cambridge, MA: Harvard University Press.
Miller, G. (2005). Neuroscience: The dark side of glia. Science, 308 (5723), 778–781.
Münte, T. F., Altenmüller, E., & Jäncke, L. (2002). The musician’s brain as a model of neuroplasticity. Nature Reviews Neuroscience, 3(6), 473–478.
Peters, M., Reimers, S., & Manning, J. T. (2006). Hand preference for writing and associations with selected demographic and behavioral variables in 255,100 subjects: The BBC Internet study. Brain and Cognition, 62(2), 177–189.
Soroker, N., Kasher, A., Giora, R., Batori, G., Corn, C., Gil, M., & Zaidel, E. (2005). Processing of basic speech acts following localized brain damage: A new light on the neuroanatomy of language. Brain and Cognition, 57(2), 214–217.
Springer, S. P., & Deutsch, G. (1998). Left brain, right brain: Perspectives from cognitive neuroscience (5th ed.). A series of books in psychology. New York, NY: W. H. Freeman/Times Books/Henry Holt & Co.
Thiel, A., Habedank, B., Herholz, K., Kessler, J., Winhuisen, L., Haupt, W. F., & Heiss, W. D. (2006). From the left to the right: How the brain compensates progressive loss of language function. Brain and Language, 98(1), 57–65.
Van Praag, H., Zhao, X., Gage, F. H., & Gazzaniga, M. S. (2004). Neurogenesis in the adult mammalian brain. In The cognitive neurosciences (3rd ed., pp. 127–137). Cambridge, MA: MIT Press.
Attributions
Content, including figures, adapted by Kenneth A. Koenigshofer, Ph.D., from Introduction to Psychology (Saylor Foundation), Chapter 3. https://saylordotorg.github.io/text_...r-thought.html; license (CC BY-NC-SA 3.0) https://creativecommons.org/licenses/by-nc-sa/3.0/. Some material on functions of cortical areas was added by Kenneth A. Koenigshofer.


