18.13: Chapter 14- The Brain and Evolution of Cognition
- Page ID
- 113224
This page is a draft and under active development. Please forward any questions, comments, and/or feedback to the ASCCC OERI (oeri@asccc.org).
1. Briefly describe two general approaches to human cognition and its evolution: continuity vs. qualitative gap
2. List and briefly describe the theories of the evolution of human intelligence discussed in this module
3. Briefly explain the possible role of cooking in human brain evolution
4. List the brain areas associated most closely with cognition and intelligence in humans and in several other animal groups
Overview
In this module, we examine ideas about the evolutionary origins of human cognition and intelligence. A number of approaches to this issue are discussed. Attempts to understand the relationship between human intelligence and intelligence in other species can be divided into two primary categories. One is based on the assumption of continuity between intelligence in human and non-human animals. The second assumes that there is a qualitative gap between human and non-human animal intelligence. Factors hypothesized to be important in the evolution of human intelligence include bipedalism, encephalization and increased information processing capacity, greater density of cortical neurons per unit of cortical mass, absolute number of neurons in the brain, meat-eating, cooking, complex social organization, "mental time travel," anticipation of future drive states, greater capacity for abstraction and visualization of future, ability to envision future actions and their outcomes, anticipatory cognition, tool use, connectivity between cortical areas and cortical networks, niche construction, and language.
Evolution of Cognition
by Kenneth A. Koenigshofer, Ph.D., Chaffey College
Animals use a range of mechanisms to respond to environmental change. These vary in complexity and sophistication from simple reflexes and simple forms of learning such as habituation to complex learning, reasoning and cognition. Cognition in this comparative context refers to the ability to acquire, process, and retain information to guide behavior and decision-making toward successful adaptation (van Horik & Emery, 2011).
Two different approaches to human cognition have dominated research and theory about evolution of cognition and intelligence. One assumes continuity between human and animal cognition and that differences are primarily a matter of degree. The second approach hypothesizes that there are major qualitative gaps or jumps in cognition and intelligence between humans and animals.
Gibson (2002) argues that the differences in cognition and intelligence between humans and other animals are a matter of degree and thus assumes continuity. According to Gibson, human cognition and intelligence exceeds that of other animals because of "increased information processing capacities" of the human brain due to enlargement of the human neocortex, cerebellum, and basal ganglia. Gibson proposes that increased information processing capabilities of the human brain led to development of human language, social cognition, and tool-making.
Herculano-Houzel (2012) offers anatomical data which supports continuity between human and non-human primate brains, thereby supporting Gibson's views. She argues that the human brain is a "scaled-up primate brain." Regarding the cerebral cortex specifically, she notes that "primate cortices contain many more neurons than nonprimate cortices of a similar mass" (Herculano-Houzel, et al., 2015, p. 159). This greater density of cortical neurons in primate cerebral cortex compared to other mammal species means that "primates are . . . subject to a different scaling rule, with more neurons for a given body mass compared to other mammalian clades. . . while larger bodies have neurons in the the ROB [rest of the brain] that are on average larger in proportion to the linear dimension of the body, the number of brain neurons is not dictated simply by body mass" (Herculano-Houzel, et al., 2015, p. 161). In other words, bigger bodies do not necessarily mean more brain neurons, but perhaps just bigger neurons. More specifically, humans have the largest brain and the largest number of neurons among the primates, perhaps as many as three times the number of neurons found in gorillas and orangutans (which have the next largest living primate brains after humans), yet gorillas can grow to be three times the body size of humans. This larger body size has not resulted in greater numbers of brain neurons in gorillas.
Why did human brains become so much larger, with so many more neurons, than expected for their body size (i.e. greater encephalization) when compared to other primates, and mammals in general? To answer this question, we need to consider the large amount of metabolic energy that a large brain with a large number of neurons requires. The energy required to run a brain increases as a function of the number of neurons that make up the brain. Brains with a lot of neurons require a lot of metabolic energy. Without sufficient energy supplies from the food available to a species, the number of brain neurons can only increase to a limit set by the available metabolic energy gained by feeding. Eating meat is one way to obtain more calories from the environment, and eating cooked meat is even better. Given that "the energetic cost of the brain is a linear function of its numbers of neurons," Fonseca-Azevedo and Herculano-Houzel (2012, p. 18571) argue that "metabolic limitations that result from the number of hours available for feeding and the low caloric yield of raw foods impose a tradeoff between body size and number of brain neurons [in non-human primates] . . . This limitation was probably overcome in Homo erectus with the shift to a cooked diet. Absent the requirement to spend most available hours of the day feeding, the combination of newly freed time and a large number of brain neurons affordable on a cooked diet may thus have been a major positive driving force to the rapid increase in brain size in human evolution." This argument is based on the observation that cooked foods are easier to digest and thus require less metabolic energy to process, leaving more usable energy after digestion compared to uncooked food. The use of fire and the invention of cooking, especially of meat, a concentrated source of metabolic energy (see below), by our ancient ancestors overcame the metabolic limitations that constrained brain size in other primates. This cultural innovation set the stage for larger relative brain size and greater number of brain neurons in human evolution beginning with Homo erectus, the oldest member of genus Homo for which evidence of the use of fire for cooking has been found. In short, eating meat, especially cooked meat, provided the metabolic energy required to support bigger brains with larger numbers of neurons, freeing hominid brain evolution beginning with Homo erectus from the metabolic limitations that prevented increased encephalization in other primates and in mammals in general.
Lakatos and Janka (2008) propose that upright posture and bipedalism in human ancestors freed the hands for evolution of improved manual dexterity leading to tool-making. Weapons improved the hunting of animals. Eating meat, as just discussed, provided more calories and fat in the diet making possible the evolution of larger brains which require large amounts of caloric energy (modern human brains comprise 2% of body weight but consume 20% of the body's metabolic energy). The human brain tripled in size over 3.5 million years from an average of 450 cm3 in Australopithecinae to 1350 cm3 in Homo sapiens. The appearance and stabilization of the FOXP2 gene (see last module in this chapter) set the stage for human language, which, according to Lakatos and Janka (p. 220), was the "basis for abrupt evolution of human intelligence," and coincided with the first appearance and the rapid spread of Homo sapiens over the Earth. They point out that the "level of intelligence is related anatomically to the number of cortical neurons and physiologically to the speed of conductivity of neural pathways, the latter being dependent on the degree of myelinization" (p.220). In other words, both the number of neurons and the amount of myelin on axons in a brain (the more myelinated axons, the faster the conduction speeds between neurons and the faster and more efficient information processing becomes, presumably increasing intelligence. In addition, they claim that language provided more effective communication among cooperating hunters and food-gatherers, requiring less energy to achieve effective cooperation, thereby providing competitive advantage in obtaining food. They also suggest that better mental skills, favored in sexual competition for mates, helped stabilize "cleverness" genes. They support the view that major qualitative changes occurred during evolution of human cognition. On their view, evolution of language was the basic condition required for conscious thinking, creating a qualitative change in human cognition which made humans and human cognition different from all other species.
Another hypothesis is the "social brain" theory of the evolution of human cognition. This stems from the observation that primate brains are roughly twice the size expected based on body size, compared to other mammals, and that the size of cerebral cortex is correlated with the size of the primate social group characteristic of a species (Barrett & Henzi, 2005). The basic idea is that living in groups created local competition among members of the social group for scarce resources which would lead to the evolution of cognitive skills to out-wit one's competitors in the social group. Increased cognitive skills would select for even greater cognitive skills because of the increased competition. However, this view of competition among individuals as the driving force in the evolution of primate cognition doesn't take into account mounting evidence that much of primate social behavior, especially in humans, involves altruistic behavior including cooperation, sharing, trade, group defense, and other prosocial behaviors which can evolve when members of a group are inter-dependent for survival and reproduction of viable offspring. This may occur through kin selection (see previous section on kin selection in Chapter 3) or group selection wherein natural selection acts on the group as a whole (Barrett & Henzi, 2005). Nevertheless, social life does involve cognitive skills not required in non-social animals, although monkeys may use less demanding cognitive skills than do apes in their social interactions. A major problem with the social brain hypothesis as an explanation for the evolution of cognition is that the argument "can sometimes appear circular: primates have large brains because their social lives are cognitively demanding, and their lives are cognitively demanding because they have large brains that allow them to produce more complex forms of social behavior" (Barrett & Henzi, 2005).
Reader, et al. (2011) note that there are conflicting hypotheses regarding the nature and structure of primate cognition. One view is that primate cognition is divided into specialized, independently evolving modules ("massive modularity"), each of which processes information using "rules" specific to solution of a specific class of adaptive problem--such as mate selection, or predator avoidance, or securing and maintaining status in one's social group, and so on. The opposite of this modular view of cognition is that primate intelligence is a single general process, attributable to a single 'general intelligence' factor, g, the evolutionary origin of which remains controversial (see Koenigshofer, 2017). Reader, et al. (2011) collected ecologically relevant measures of cognition including reported incidences of behavioral innovation, social learning, tool use, extractive foraging and tactical deception, in 62 primate species. All measures exhibited strong positive associations, after statistically controlling for multiple potential confounds, creating a highly correlated composite of cognitive traits which they took as a species-level measure of general intelligence, which they called, "primate g(S)." They argue that primate g(S) suggests that "social, technical and ecological abilities have coevolved in primates, indicative of an across-species general intelligence that includes elements of cultural intelligence," such as social learning ability and tool use. They also reported that primate g(S) correlated "with both brain volume and captive learning performance measures." These researchers conclude that their "findings question the independence of cognitive traits and do not support 'massive modularity' in primate cognition, nor an exclusively social model of primate intelligence" given that no relationship between social group size (complexity) and cognitive performance in lab tests was found. This suggests that social complexity as measured by group size may not have been as important a driver of the evolution of primate general intelligence as is widely believed. "High general intelligence has independently evolved at least four times, with convergent evolution in capuchins, baboons, macaques and great apes" (Reader, Hager, & Laland, 2011, p. 1017). "The strong correlation between distinct measures of primate cognitive performance is strikingly evocative of the correlations in performance on different IQ tests observed in humans. A possible explanation for this correspondence is that the g factor reported in humans reflects underlying general processes that evolved in common ancestors and are thus shared in our extant primate relatives" (Reader, Hager, & Laland, 2011, p. 1024). This view is consistent with arguments made in module 14.2 that general intelligence is found in many non-human animals as well as in humans (Koenigshofer, 2017).
Another intriguing hypothesis about the evolution of cognition and the gap or discontinuity between human cognition and cognition in other animals is the concept of mental time travel. "Mental time travel comprises the mental reconstruction of personal events from the past (episodic memory) and the mental construction of possible events in the future" (Suddendorf, Addis, & Corballis, 2011, p. 344). This projection of the self into future-time using visual imagery (i.e. imagination) was discussed as an important component of general intelligence (Koenigshofer, 2017) in module 14.2. According to Suddendorf and colleagues, the ability to "time travel" to the past doesn't occur in human children until about three and a half years old, and is associated with episodic memory, a type of autobiographical memory dependent upon functioning of the prefrontal cortex involving understanding that the memory is of an experience of a past self. This awareness of "pastness" may be beyond the capability of most animals, particularly if they do not have a conscious sense of self. Furthermore, mental time travel requires ability to dissociate from one's current state of mind to generate a personal memory or a projection into possible future states of the world (imagination again). This involves so-called "executive functions" of the frontal lobe. "'Executive function' is an umbrella term for the mental operations which enable an individual to disengage from the immediate context in order to guide behavior by reference to mental models and future goals" (Hughes, Russel, & Robbins, 1994, p. 477, as referenced in Suddendorf, et al., 2011). As was discussed in earlier modules, this ability requires the prefrontal cortex. Persons with autism and those with prefrontal damage have difficulty with executive functions. This cognitive impairment is revealed by inability to switch behavior previously employed when changes in the task require, and also by apparent difficulty in using mental models as evidenced by deficits in planning future behavior. Although animals in general certainly anticipate future in various ways, including implicit learning such as conditioning, whether they construct mental models of future is for most species an open question; however, for chimpanzees at least, evidence from Kohler's classic studies showed sudden insight by chimpanzees in problem solving implying "constructive thought with an eye to the future solution of a problem" suggesting mental time travel to the future in these animals. Studies by Jane Goodall of chimpanzee tool-making "also suggest flexible forethought. For example, the chimpanzees at Gombe [in Tanzania] manufactured pointed tools from sticks at one place to use them later for termite fishing at another place that was out of sight (Goodall, 1986)." Furthermore, chimpanzees have been observed to transport stones for cracking open nuts from one region where stones are found to another feeding location when stones are not found. This shows ability to imagine future and to engage in current behavior that anticipates and plans for an expected future.
However, one hypothesis by Bischof (1978, 1985) is that non-human animals cannot anticipate future needs or drive states and so are bound by their present motivational state. For example, a full-bellied lion is no threat to passing zebra, but a full-bellied human may be (Suddendorf, Addis, & Corballis, 2011, p. 349). This difference may reflect a trend in evolution. "Bischof (1985) suggests that, in the course of evolution, there was a progressively increasing gap between drive and action. Great apes display quite extensive gaps; they can postpone the immediate enactment of their current drive, and make plans to receive gratification at a later point in time." The role of human anticipatory cognition is not only central to human behavior but shapes human society as well (Suddendorf, Addis, & Corballis, 2011, p. 351). For example, we grow, harvest, store, and ship food for future use using all sorts of complex behavioral practices oriented to the future use of the food. We have complex legal systems that are based on the idea that current punishment for a past crime will prevent future crime. We have invented nuclear arsenals ready for launch based on the assumption that such weapons will prevent future attacks on us. We have the institution of marriage which we anticipate will help satisfy our needs for emotional and financial security (including needs for shelter and food), for reproduction of offspring, and for future sexual gratification.
Osvath and Gärdenfors (2005, p. 1), following Bischof's hypothesis, state "Anticipatory cognition, that is, the ability to mentally represent future needs, is a uniquely human trait that has arisen along the hominid line . . . [resulting in] stone tool manufacture, . . . transports over long ranges of tools as well as food and of the use of accumulation spots. . . .[T]his niche promoted the selection for anticipatory cognition, in particular planning for future goals. Once established, anticipatory cognition opened up for further cultural developments, such as long ranging migration, division of labor, and advanced co-operation and communication, all of which one finds evidence for in Homo ergaster/erectus. . . . A distinctive feature of human thinking that contrasts with the cognition of other primates is our capacity to form mental representations of the distant future. . . . planning in primates and other animals suggest that they can only plan for present needs (this is dubbed the Bischof-Kohler hypothesis) . . . "
The evolution of anticipatory cognition resulted in "niche construction," a term from evolutionary biology. "Niche construction can be defined as the systematic changes that organisms bring about in their environments (Day, Laland & Odling-Smee, 2003). A wide variety of organisms construct parts of their environment: spiders making webs, birds building nests, beavers constructing dams and plants altering their chemical surroundings" (Osvath and Gärdenfors (2005, p. 2). Of particular interest is the fact that niche construction "can actually change the evolutionary dynamics" by altering selection pressures leading to "unusual evolutionary dynamics, . . . [including] ecological inheritance. This is characterized by offspring inheriting the altered ecology from its ancestors – an ecology with its own selective pressures. Implementing niche construction in evolutionary theory involves two kinds of modifying processes, natural selection and niche construction, as well as two kinds of descent, genetic and ecological inheritance (Day, Laland & Odling-Smee, 2003)" (Osvath and Gärdenfors (2005, p. 2). According to Osvath and Gärdenfors (2005, p.1), "the cultural niche that was created by the use of Oldowan [stone] tools, including transport of tools and carcasses (Plummer, 2004), has led to a selection for anticipatory cognition, and in particular anticipatory planning." These events produced a qualitative gap in cognitive evolution between humans and apes--our capacity for representation of the distant future, including planning for future needs, capacities which appear to be absent in other animals including our nearest primate relatives, the great apes.
"The ability to envision various actions and their consequences is a necessary requirement for an animal to be capable of planning. . . An organism is planning its actions if it has . . . representations of (1) goal and start situations, (2) sequences of actions, and (3) the outcomes of actions. . . . The hominids have presumably been the most cultural dependent species ever and thus the most powerful and flexible niche constructors (to various degrees, of course, and with Homo sapiens at the present extreme). A consequence of this is that the hominids should exhibit great evolutionary resistance to changes in the natural environment, but also be capable of far-reaching evolutionary change caused by some ground-breaking cultural innovation (Laland, Odling-Smee & Feldman, 2000). The key point is that some of our major cognitive adaptations might not only be a result of new demands from a changing natural environment, but to a larger degree emerge from a highly constructed environment, including both social and artefactual elements. It is not sufficient to look at habitat, climate and other natural ecological factors, when trying to understand how humans evolved" (Osvath and Gärdenfors (2005, p. 2). Niche construction must also be considered, and Osvath and Gärdenfors argue that niche construction played a large role in human cognitive evolution.
They explain further: "Culture is the most forceful medium in niche construction, because culturally transmitted traits spread much more rapidly than genetically transmitted traits do. On one hand, culture can create a very strong selection and thereby increase the rate of evolutionary changes. On the other hand, culture can buffer out particular natural selection pressures (Laland, Odling-Smee & Feldman, 2000). A cultural innovation, constituting a form of niche construction, can create a behavioural drive that accelerates evolution. The same innovation can create a niche that blocks natural selective pressures. For example, humans live in cold climates without having any major biological adaptations to cold, because they rely on the cultural niche created by clothes and external heat sources. The consequence is that the naked human body does not exhibit cold adapted traits to the extent that would be expected from a mammal living in a cold climate. . . The general thesis is that the hominids themselves created a great deal of the selective pressures that eventually made us human."
Figure \(\PageIndex{1}\): Human niche construction: from stone tools to skyscrapers. (Images from Wikimedia Commons. (Left) File:Bokol Dora Stone Tool (cropped).jpg; https://commons.wikimedia.org/wiki/F..._(cropped).jpg; by David R. Braun; licensed under the Creative Commons Attribution-Share Alike 2.0 Generic license. (Right) File:Nakanoshima Skyscrapers in 201705 002.jpg; https://commons.wikimedia.org/wiki/F...201705_002.jpg; by Mc681; licensed under the Creative Commons Attribution-Share Alike 4.0 International license.
Another hypothesis about the difference between human and animal cognition emphasizes capacities for abstraction. Ability for high levels of abstraction in causal and similarity relations (Penn et al., 2008) and in covariation detection (Wasserman, 1993b) may be central to the phylogenetically unprecedented achievements of the human intellect (Koenigshofer, 2017), including niche construction. In a review of the comparative literature, Penn et al. (2008) find “no compelling evidence” that any nonhuman animal can reason about the “relation between relations,” something that humans do readily. Neither do they find convincing evidence for analogical reasoning (dependent upon judgments of abstract similarities across domains) in nonhuman animals. Children readily understand the similarity between a dog and its dog house and a bird and its nest. Consistent with Penn et al. (2008), Walker and Gopnik (2014) report that although human toddlers readily detect abstract relational causality quickly with only a few training trials, non-human primates have great difficulty with similar tasks even after hundreds of trials. Penn et al. (2008, p. 123) believe that what distinguishes human cognition from that in non-human animals is “the ability to reason about higher-order relations,” and that this capacity for high levels of abstraction “subserves a wide variety of distinctively human capabilities.”
As discussed in an earlier module, research in molecular genetics suggests one possible explanation for how human ability for high levels of abstraction may have come about during the course of evolution. Pollard (2009) comparing human and chimpanzee genomes found “massive mutations” in humans in the “DNA switches” controlling size and complexity of cerebral cortex, extending the period of prenatal cell division in human cerebral cortex by several days compared to our closest primate relatives. Research using artificial neural networks suggests that increasing cortical complexity leads to sudden leaps in ability for abstraction and rule-like understanding of general principles (Clark, 1993), a hallmark of high intelligence. Findings by Penn et al. (2008) strongly suggest that superior ability for abstraction of relational information may be the key component explaining differences in general intelligence between humans and nonhuman animals. These abilities may involve anterior dorsolateral prefrontal cortex in humans (Kroger et al., 2002; Reeber et al., 1998; see Koenigshofer, 2017).
Frontal cortex is found in other mammals as well, so the question remains as to why the human brain with its frontal lobe has capabilities which exceed those of all other animals. In this regard, Roth and Dicke (2012) note "Primates are, on average, more intelligent than other mammals, with great apes and finally humans on top. They generally have larger brains and cortices, and because of higher relative cortex volume and neuron packing density (NPD), they have much more cortical neurons than other mammalian taxa with the same brain size. Likewise, information processing capacity is generally higher in primates due to short interneuronal distance and high axonal conduction velocity [see discussion above of findings by Herculano-Houzel, et al., 2015, and Lakatos and Janka, 2008]. Across primate taxa, differences in intelligence correlate best with differences in number of cortical neurons and synapses plus information processing speed. The human brain stands out by having a large cortical volume with relatively high NPD, high conduction velocity, and high cortical parcellation. All aspects of human intelligence are present at least in rudimentary form in nonhuman primates or some mammals or vertebrates except syntactical language. The latter can be regarded as a very potent 'intelligence amplifier.'" This last point is related to human ability to share knowledge and ideas facilitating cultural transmission of learned information and cultural evolution.
Another important finding involves the connectivity between cortical areas. For example, prefrontal white matter volume is disproportionately larger in humans than in other primates (Schoenemann, et al., 2005). However, this finding might be related to more gyri in the frontal cortex of humans compared to other primates. Nevertheless, these observations may reflect "connections between cortical areas capable of relaying and processing information at greater speeds, facilitating complex cognitive function" (Schenker, et al., 2005, p.564).
Williams (2020) emphasizes the role of cognitive interactions within human groups to account for the cognitive achievements of humans compared to other animals. According to Williams, General Collective Intelligence (GCI) can be defined as a system that organizes groups into a single collective cognition with the potential for vastly greater general problem solving ability than that of any individual in the group. According to Williams, GCI represents a phase transition in human intelligence. When humans collectively put their cognitive capacities and activity together to solve a particular problem a collective intelligence emerges which is far more powerful than the cognitive abilities of any single individual. Communication and social cooperation between individual humans "putting their heads together" facilitated by cultural innovations for sharing information and ideas, within and across generations (such as writing, the internet, scientific organizations, research universities and other group cognitive efforts), create a kind of "superintelligence" not found in any other species. The powerful adaptive effects of collaborative problem solving combined with cultural transmission of learned information and ideas from generation to generation, built upon by each succeeding generation, is essential to the cognitive achievements which make us so different from other species. While other species such as dogs, apes, killer whales, dolphins, elephants and so on are quite intelligent, none has created an international space station or flown robots to Mars.
Nevertheless, cognition and intelligence have evolved in a wide variety of species. "Within the animal kingdom, complex brains and high intelligence have evolved several to many times independently, e.g. among ecdysozoans in some groups of insects (e.g. blattoid, dipteran, hymenopteran taxa, e.g. bees, wasps), among lophotrochozoans in octopodid molluscs (e.g. octopus), among vertebrates in teleosts (e.g. cichlid fish), corvid and psittacid birds (e.g. ravens, crows, parrots), and cetaceans e.g. whales, dolphins, porpoise), elephants and primates. High levels of intelligence are invariantly bound to multimodal centres such as the mushroom bodies in insects, the vertical lobe in octopodids, the pallium in birds and the cerebral cortex in primates, all of which contain highly ordered associative neuronal networks. The driving forces for high intelligence may vary among the mentioned taxa, e.g. needs for spatial learning and foraging strategies in insects and cephalopods, for social learning in cichlids, instrumental learning and spatial orientation in birds and social as well as instrumental learning in primates" (Roth, 2015, p. 0049). According to Roth, high intelligence, which evolved in a large variety of species, is dependent not only upon factors identified by others such as number of brain neurons, their packing density, myelination and speed of processing, but also upon the presence of "multimodal centers" where processing events from multiple sensory modalities can be integrated into whole object and whole event representations. Keep in mind, that as was discussed in Module 14.2, cognition and intelligence in any species are part of the guidance systems that direct behavior toward successful adaptation in response to the challenges and opportunities presented by the environment. In the human case, the control of fire for cooking food, especially meat, permitting larger numbers of brain neurons in Homo erectus and in the members of the Homo genus which followed appears, to have been critical for setting hominids on the path to brain expansion and increasingly sophisticated cognition. But the fossil record shows us that a large brain was a prominent feature not only of ancient Homo sapiens in Africa but also of the Neanderthals in Europe, east Asia, and the Middle East. Although, Neanderthals had a larger visual cortex than modern humans, accounting for some of their enlarged cranial capacity, their frontal lobes, associated with human intelligence, were approximately the same size as the frontal lobes of modern humans, suggesting the possibility of high intelligence in the Neanderthals.
Differentiation of the Frontal Cortex
The role of the frontal cortex in human intelligence cannot assume a uniform set of regions comprising this cortex. Instead research shows that the frontal cortex is differentiated into regions specialized for different functional roles. For example, Schenker et al. (2005, p. 547-548) note that "this large cortical territory comprises anatomical subdivisions with rather distinct functional attributes. The dorsal sector of the frontal lobe is involved in perception, response selection, working memory, and problem solving (Owen, 1997; Bechara et al., 1998; Petrides, 2000; Pochon et al., 2001). It also includes territories activated during language production in humans (Foundas, 2001) and most of the primary motor and premotor cortices (Zilles et al., 1995). The mesial frontal cortex, including the anterior cingulate, is important for processing emotional stimuli and for production of affective responses (Cummings, 1993; Rezai et al., 1993), planning and initiation of voluntary motor sequences (Tanji and Mushiake, 1996), theory of mind (Fletcher et al., 1995; Gallagher et al., 2000; Stuss et al.,2001), and attention management (Carter et al., 1999; Dagher et al., 1999). The orbital frontal cortex is known to be involved in the evaluation of actions based on emotional reinforcers (Damasio, 1994; Stone et al., 1998; Rolls, 2000)." It is not unreasonable to speculate that these functions of the orbital frontal cortex may play a role in human ability to plan behavior for future needs, which according to the Bischof's hypothesis, discussed above, is a key feature of human cognition setting it apart from cognition in other primates.
Evolution of Increased Brain Size in Neanderthals and Early Humans
As discussed above, theorists have hypothesized many different factors to account for the expansion of the human brain and increases in intelligence. Fonseca-Azevedo and Herculano-Houzel (2012) hypothesized that a cooked diet was key to greater encephalization (larger brain and more neurons)--humans have the largest brain and the largest number of neurons among the living primates, perhaps as many as three times the number of neurons found in gorillas and orangutans. Lakatos and Janka (2008) proposed that upright posture and bipedalism in human ancestors freed the hands for improved tool-making and production of weapons which improved hunting, providing a richer diet of meat capable of sustaining evolution of larger brains. Also recall the "social brain" hypothesis that the evolution of intelligence and cognition in humans was stimulated by increased information processing required by complex social interactions.
Evolution of Brain Size and Intelligence in Neanderthals
Now consider the Neanderthals whose Mousterian manufacturing indicates higher levels of conceptualization. Neanderthal fossil skulls show brain size equal to or slightly larger than that of modern humans (see figure below). The size of frontal cortex in humans and Neanderthals is essentially the same. Considering their large brain size and frontal lobes comparable to those of modern humans, some anthropologists suggest that Neanderthal cognitive and social capacities were not significantly different from that in humans 30,000 years ago (Hayden, 2012). Given these facts, we may wonder to what extent the above hypotheses about the evolution of human intelligence might also apply to Neanderthals. Spear points at Neanderthal sites certainly suggest hunting and meat-eating by Neanderthals. Neanderthals also cooked their food (Hayden, 2012; Henry, 2017). There is evidence that Neanderthals lived in groups (although perhaps smaller than humans of 40,000 years ago) and may have had complex social interactions (Duveau et al., 2019; Hayden, 2012). Could any or all of these factors, hypothesized as catalysts for the evolution of complex cognition in humans, have also been catalysts for the evolution of greater brain size and higher intelligence in Neanderthals?
Cognitive Competition between Early Humans and Neanderthals in Eurasia
Another intriguing question involves possible competition between early humans and Neanderthals. Some researchers suggest that competition from humans migrating to Europe and Asia from Africa eventually caused the extinction of the Neanderthals. However, another possible effect of such competition may have been beneficial for both species. Could competition between early humans and Neanderthals have been a factor stimulating evolution of higher intelligence in both species, with humans eventually winning out? This hypothesis is weakened by the fact that no fossils of Neanderthals have ever been found in Africa, but only in Europe, the Middle East, and parts of east Asia. Without such fossil finds of Neanderthals in Africa we must assume that no interactions in Africa between early Homo species and Neanderthals were possible, eliminating competition between the two species as a potential cause of increased brain size and intelligence in early humans in Africa. But what about potential competition between the two species in Eurasia? Was contact between the two species frequent and sustained enough to create significant cognitive competition between them which might have influenced their brain evolution?
It is now generally accepted by scientists that Neanderthals and modern humans coexisted for tens of thousands of years, Neanderthals having become extinct only some 40,000 years ago, while the origin of modern humans is thought to have taken place some 200,000 to 300,000 years ago and human migration out of Africa to Europe and Asia is thought to have occurred about 70,000 to 100,000 years ago. Furthermore, about 1-4% of human DNA is Neanderthal in origin indicating interbreeding, suggesting significant contact between Neanderthals and humans in several places outside of Africa and during several periods between 40,000 and 100,000 years ago (Alex, 2018). Anthropological evidence suggests that Neanderthals were cognitively advanced. For example, they made stone tools for specialized tasks. These tools, found in a cave in France, were made using a manufacturing technique (dubbed Mousterian by experts) characterized by preparing the core of raw stone material from which flakes were struck and then worked. Sharper tools with a finer edge can be produced using this technique. Neanderthals shaped these flakes into tools like scrapers, blades, and projectile points--specifically spear points. In fact, at Neanderthal caves sites in the Middle East, there are a higher percentage of spear points found than at neighboring Homo sapiens sites. Mousterian tools are a technological advance, taking a high degree of conceptualization and knowledge of the properties of the stone (see section on Material Culture in this textbook).
Were Neanderthals smart enough to provide cognitive competition for early humans? Human and Neanderthal DNA is 99.8% the same including genes important for brain expansion and language (Alex, 2018). As already mentioned, measurements of endocranial volume of Neanderthal skulls indicate that Neanderthals possessed a brain similar in size or even slightly larger than the brains of recent humans, with a larger visual cortex in Neanderthals, but a larger cerebellum in humans (Alex, 2018; Kochiyama, et al., 2018). But was any potential competition between Neanderthals and ancient humans sustained long enough to impact brain evolution? As discussed, we do know that the interactions between the two species were sufficient for some interbreeding between them, at least in Eurasia. Whether interactions between the two species led to cognitive competition between the two species that was sufficient to affect brain evolution of one or both species is unknown, but remains an intriguing question. However, one thing we do know is that most of the encephalization of the two species was already complete before early humans left Africa and therefore before any interactions could have taken place between them.
Sometime between 520,000 and 630,000 years ago the human and Neanderthal lines diverged from a common ancestor (Homo heidelbergensis; see figure below) and took separate evolutionary paths. Those who migrated to Europe eventually evolved into Neanderthals, while those in Africa gave rise to modern humans. These early humans later migrated out of Africa to Europe and other regions of the Earth. By 150,000 years ago both species had similar average brain size of about 1400 cubic centimeters, three times larger than the brains of modern day chimpanzees, and they also had very similar within-species variation in cranial capacity. The average brain volume of Neanderthals of 1410 cubic centimeters actually exceeds the 1349 cubic centimeter average of modern humans (Alex, 2018). These events appear to have occurred before any interactions between the two species, suggesting that the bulk of brain evolution in both took place before any encounters between them, further weakening the mutual competition hypothesis discussed above as a significant cause of the encephalization that occurred in the evolution of early humans and Neanderthals after they diverged from Homo heidelbergensis.
What accounts for the similar large brain size in both Neanderthals and humans? Were the genetics for large brain size present in the common ancestor of both before the two lines diverged? Or, alternatively, did the similar encephalization in the two species result from convergent evolution? Common genetics appears to be at least one important factor. Homo heidelbergensis is thought to be the common ancestor of Homo neanderthalensis in Europe and Homo sapiens in Africa (see section on Human Evolution in Chapter 3). The trend toward larger brain size was already evident in Homo habilis and Homo erectus, predecessors of Homo heidelbergensis, suggesting that the increase in encephalization in Neanderthals and Homo sapiens was a continuation of the trend toward encephalization already present in the genus Homo. The figure below from section 3.7 shows these evolutionary relationships.
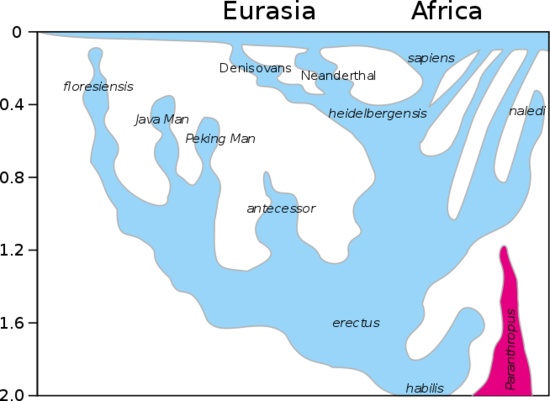
Figure \(\PageIndex{2}\): Evolutionary relationships among hominins. Note the divergence of Neanderthal and sapiens (the human line) from heidelbergensis, which evolved from habilis and erectus. (Image from Wikimedia Commons; File:Homo lineage 2017update.svg; https://commons.wikimedia.org/wiki/F...2017update.svg; by User:Conquistador, User:Dbachmann; licensed under the Creative Commons Attribution-Share Alike 4.0 International license).
One hypothesis about the disappearance of the Neanderthals was that competition from early humans led to their extinction some 30,000 to 40,000 years ago (Gilpin, et al., 2016). However, as suggested above, is it possible that competition between Neanderthals and humans in Europe, south Asia, and the Middle East (where fossils of early humans and Neanderthals have been found) might have played some role in stimulating the evolution of larger, smarter brains in both species? In the figure below, note the larger skull of the Neanderthal compared to the skull of a modern human. Many of the species of the Homo genus coexisted with one another and could have interacted with one another. Perhaps competition between various Homo species played some role in the trend toward greater encephalization seen in the genus Homo beginning with species which diverged from Homo erectus. However, this hypothesis requires evidence of significant and sustained competition between these species, and that evidence has remained elusive. Perhaps future fossil finds will shed light on this intriguing possibility. On the other hand, small populations of these species may have precluded significant overlap of their ranges and reduced the chances of significant interaction and sustained competition among them.
Figure \(\PageIndex{3}\): Comparison of Modern Human and Neanderthal skulls from the Cleveland Museum of Natural History. The cranial space in Neanderthal reveals a brain size slightly larger than the brain size of modern humans. This and other anthropological evidence suggests the possibility of significant competition between the two species, perhaps catalyzing the evolution of cognition in both. (Image and first sentence in caption from Wikimedia Commons, remainder of caption by Kenneth Koenigshofer, PhD, Chaffey College; File:Sapiens neanderthal comparison en.png; https://commons.wikimedia.org/wiki/F...parison_en.png; by hairymuseummatt (original photo), KaterBegemot (derivative work); licensed under the Creative Commons Attribution-Share Alike 2.0 Generic license).
An Integrative Model of the Evolution and Structure of Intelligence
A significant controversy among theorists is whether the human mind is modular or general purpose. The older model of the mind, originating with the British Empiricist philosophers of the 17th and 18th centuries and adopted by early behaviorists, sees the brain as a general purpose processor handling all kinds of adaptive problems without need for any specialized processing brain modules. An alternative model favored by many evolutionary psychologists such as Cosmides and Tooby at the University of California, Santa Barbara, is that the human mind is highly modular in nature, consisting of large numbers of specialized, domain-specific (specific to specific problem type) information processing modules which operate more or less independently of one another. Such modules or mini-computers are hypothesized to be specialized by evolution to operate on different types of information to achieve solutions to different types of adaptive problems. Although many evolutionary psychologists favor this modular view of the mind and brain, composed of special purpose brain modules, many psychologists disagree with this view, in part because it leaves out general intelligence and the flexibility in problem solving that seems to require general problem solving capabilities.
One solution that integrates both views is a model of the human brain that incorporates both types of processing mechanisms--some brain circuits specialized to solve specific adaptive problems such as mate selection, predator avoidance, and so on, and, in addition, brain mechanisms of general intelligence. Many evolutionary psychologists do not dispute the idea that some form of general intelligence exists; the problem is that they have been unable to understand how general intelligence, capable of solving noncurrent "novel problems," could have even evolved (Cosmides & Tooby, 2002; Kanasawa, 2004, 2010). This is because problems that are truly novel have not happened before and therefore have not been recurrent over many generations. This creates a theoretical problem because natural selection can only work on selection criteria consistently present generation after generation.
However, this problem has been solved. An analysis of the structure of events, including adaptive problems, reveals that every event contains two components: 1) abstract relational properties, such as cause-effect, similarity, and predictive relations, which have been consistently present in the environment generation after generation, and 2) specific event details which are idiosyncratic, nonrecurrent, perhaps even truly novel, and therefore transient and confined to a single generation (Koenigshofer, 2017). Any adaptive problem is a mixture of these two elements. Although event details which are idiosyncratic to specific situations encountered by different individuals may be unique and novel, and thus highly variable across generations, nevertheless the relational properties such as cause-effect exist across situations and adaptive problems generation after generation.
Because of the repetition of such relational properties (causal, similarity, and predictive relations) over countless generations in countless events, adaptive problems, and adaptive opportunities, natural selection has utilized these relational regularities of the world as selection criteria for evolution of important components of brain organization and function. As a consequence, information about these relational properties of the world has been genetically incorporated into the innate organization of the brains of many species, including our own (Koenigshofer, 2017). This innate knowledge about these relational properties of the world forms a set of cognitive instincts that collectively make up what psychologists call general intelligence (Koenigshofer, 2017), the g-factor first discovered by Spearman in the early 1900s.
On this view, novel problems are only novel in their details, but constant or invariant across generations in their abstract relational structures. It is the constancy of these relational structures over generations that provided the regularities across generations which acted as selection criteria for the evolution of innate knowledge about these abstract, relational invariants--the key components of general intelligence. Application of this innate knowledge of general intelligence permits the solution to a near infinite variety of problems and adaptive opportunities, regular in their relational structure, but novel in their details (Koenigshofer, 2017). According to this integrative model of intelligence, many specialized modules (the modular view of intelligence) act along with general intelligence--the innate knowledge of causal, similarity, and predictive relations--to flexibly solve problems and seize adaptive opportunities, accounting for the unprecedented achievements of the human intellect. These cultural innovations by humans and their preservation over generations by cultural transmission have permitted humans each generation to draw upon the innovations and inventions of prior generations, building upon them, generation after generation, ratcheting up human adaptive success, far beyond that found in any other species.
References
Alex, B. (2018). Discover Magazine. Neanderthal Brains: Bigger, Not Necessarily Better; https://www.discovermagazine.com/pla...ssarily-better; retrieved 3/22/2022.
Barrett, L., & Henzi, P. (2005). The social nature of primate cognition. Proceedings of the Royal Society B: Biological Sciences, 272 (1575), 1865-1875.
Duveau, J., Berillon, G., Verna, C., Laisné, G., & Cliquet, D. (2019). The composition of a Neandertal social group revealed by the hominin footprints at Le Rozel (Normandy, France). Proceedings of the National Academy of Sciences, 116(39), 19409-19414.
Fonseca-Azevedo, K., & Herculano-Houzel, S. (2012). Metabolic constraint imposes tradeoff between body size and number of brain neurons in human evolution. Proceedings of the National Academy of Sciences, 109 (45), 18571-18576.
Gibson, K. R. (2002). Evolution of human intelligence: The roles of brain size and mental construction. Brain, behavior and evolution, 59 (1-2), 10-20.
Gilpin, W., Feldman, M. W., & Aoki, K. (2016). An ecocultural model predicts Neanderthal extinction through competition with modern humans. Proceedings of the National Academy of Sciences, 113 (8), 2134-2139.
Hayden, B. (2012). Neandertal social structure?. Oxford Journal of Archaeology, 31 (1), 1-26.
Henry, A. G. (2017). Neanderthal cooking and the costs of fire. Current Anthropology, 58 (S16), S329-S336.
Herculano-Houzel, S. (2012). The remarkable, yet not extraordinary, human brain as a scaled-up primate brain and its associated cost. Proceedings of the National Academy of Sciences, 109 (Supplement 1), 10661-10668.
Herculano-Houzel, S., Catania, K., Manger, P. R., & Kaas, J. H. (2015). Mammalian brains are made of these: a dataset of the numbers and densities of neuronal and nonneuronal cells in the brain of glires, primates, scandentia, eulipotyphlans, afrotherians and artiodactyls, and their relationship with body mass. Brain, Behavior and Evolution, 86(3-4), 145-163.
Kochiyama, T., Ogihara, N., Tanabe, H.C. et al. (2018). Reconstructing the Neanderthal brain using computational anatomy. Sci Rep 8, 6296. https://doi.org/10.1038/s41598-018-24331-0
Lakatos, L., & Janka, Z. (2008). Evolution of human brain and intelligence. Ideggyogyaszati szemle, 61 (7-8), 220-229.
Osvath, M., & Gärdenfors, P. (2005). Oldowan culture and the evolution of anticipatory cognition. Lund University Cognitive Studies, 122, 1-16.
Reader, S. M., Hager, Y., & Laland, K. N. (2011). The evolution of primate general and cultural intelligence. Philosophical Transactions of the Royal Society B: Biological Sciences, 366(1567), 1017-1027.
Roth, G. (2015). Convergent evolution of complex brains and high intelligence. Philosophical Transactions of the Royal Society B: Biological Sciences, 370 (1684), 20150049.
Roth, G., & Dicke, U. (2012). Evolution of the brain and intelligence in primates. Progress in brain research, 195, 413-430.Suddendorf, T., Addis, D. R., & Corballis, M. C. (2011). Mental time travel and shaping of the human mind. M. Bar, 344-354.
Schenker, N. M., Desgouttes, A. M. & Semendeferi, K. (2005) Neural connectivity and cortical substrates of cognition in hominoids. Journal of Human Evolution 49(5):547–69
Schoenemann, P. T., Sheehan, M. J. & Glotzer, L. D. (2005) Prefrontal white matter volume is disproportionately larger in humans than in other primates. Nature Neuroscience 8(2):242–52.CrossRefGoogle ScholarPubMed
van Horik, J., & Emery, N. J. (2011). Evolution of cognition. Wiley Interdisciplinary Reviews: Cognitive Science, 2(6), 621-633.
Williams, A. E. (2020). Human intelligence and general collective intelligence as phase changes in animal intelligence.
Attributions
"Evolution of Cognition" by Kenneth A. Koenigshofer, Ph.D., Chaffey College, licensed under CC BY 4.0.


